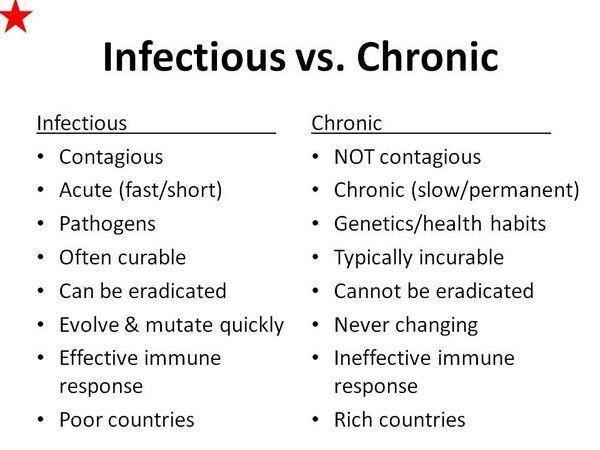
Infectious diseases comprise those illnesses that are caused by microorganisms or their products. Clinical manifestations of infection occur only when sufficient tissue injury has been inflicted directly by microbial products (e.g., endotoxins and exotoxins), or indirectly by host responses (e.g., cytokines and hydrolytic enzymes released by polymorphonuclear leukocytes). Despite the extraordinary recent advances that have occurred in therapeutics for infectious diseases, a number of basic principles should be followed to prescribe antimicrobials and vaccines is an optimal manner. This chapter addresses the broader issues of treating infectious diseases and provides a number of practical clinical examples to demonstrate rational therapeutics.
A rational therapeutic strategy in the management of proved or suspected infectious diseases must focus on the following:
- Performing a history and physical examination, and from the data obtained deciding on the infection syndrome that is the most likely explanation for the patient’s illness (e.g., endocarditis, urosepsis, pneumonia)
- Collection of the appropriate specimens to make a microbiological etiological diagnosis
- Utilizing local epidemiological data (e.g., 30% of Streptococcus pneumoniae isolates in this community are resistant to penicillin) in choosing an appropriate empirical antimicrobial regimen with which to treat your patient
- Tailoring the dose and route of administration of the antimicrobial in accordance with host factors (severity of illness, shock, renal failure, immunosuppression, etc.)
- Moving from broad-based empiric therapy to more specific, narrow-spectrum therapy once an etiologic diagnosis is available
- Monitoring response to treatment so that treatment failure and adverse drug reactions are recognized
- Assessing the risk of this infection to the community (e.g., tuberculosis, meningococcal disease in a school) and informing public health authorities when appropriate
- Assessing the opportunities for prevention of this and other infections in this patient
The importance of appropriate decisionmaking is highlighted by the fact that antimicrobials are among the most commonly prescribed drugs on a worldwide basis. Antibiotics account for one of five new and refill prescriptions each year. Furthermore, hospital purchases of antibiotics usually represent 25% to 30% of the annual drug budget for the institution. Each tertiary care hospital in the United States typically spends more than $1 million per annum on antibiotic purchases alone.
Given this tremendous utilization of antimicrobials, it is disturbing that numerous carefully performed surveys at private and university-affiliated facilities have indicated that the majority of hospitalized patients had no evidence of infection to justify antimicrobial usage or were treated with inappropriate dosage or inappropriate antimicrobials with respect to the infectious disease process. A recent study of 1529 U.S. physicians who treated 28,787 patients on an ambulatory basis found that antibiotics were frequently prescribed for viral respiratory tract infections. When these data were extrapolated nationwide, 12 million antibiotic prescriptions accounting for 21% of all antibiotic prescriptions to adults in 1992 were for treatment of viral infections.
These examples of excessive utilization and misuse of antibiotics justify ongoing educational programs related to improving usage of antimicrobials. However, educational programs alone will probably not correct excessive antibiotic misuse. In a hospital setting, requiring prior authorization (from the Infectious Disease Service) for the use of selected antimicrobials resulted in a decrease of up to 32% in the expenditures for parenteral antimicrobials in a teaching hospital.
The financial considerations for hospitals, health insurance companies, and patients related to the costs of inappropriate antibiotic therapy represent a potentially serious misuse of health care resources to the extent that compromises are made in delivery of other necessary patient care services. If physicians do not recognize and respond to this problem, bureaucratic policies regarding utilization may be instituted. However, the most important consequence of the misuse of antibiotics is the widespread emergence of antibiotic resistance and the attendant loss of effective antibiotics to treat life-threatening infections.
Principles
Overuse and misuse of antibiotics not only waste health carehealth care dollars, but also have a hidden cost of inducing antibiotic resistance in important pathogens, making serious infections more difficult to treat.
Epidemiologic and virulence factors in infectious diseases
Documentation of infection
Antimicrobial therapy: general principles
Toxicity of Antimicrobial Therapy
Nucleoside analogs
Nonnucleoside reverse transcriptase inhibitors
This group of agents, first described in 1990, acts by binding to the reverse transcriptase enzyme at a location below the catalytic site, resulting in its inactivation. These drugs, unlike the nucleoside analogs, do not require phosphorylation to become active. They are highly selective against human immunodeficiency virus-1 and very potent in vitro. Their major drawback is the rapidity with which resistance (that is considered cross-resistant for all Nonnucleoside reverse transcriptase inhibitors) occurs. A major advantage of two agents in this class is the ability to prescribe once daily dosing. Several studies with Nonnucleoside reverse transcriptase inhibitors have been completed. Although surrogate marker benefit has been shown when used in combination regimes, none of the studies have yet demonstrated an effect on disease progression. They have been generally well tolerated, the major problem being the rash associated with two of them.
Nevirapine was the first of the Nonnucleoside reverse transcriptase inhibitors to be licensed. It is synergistic in vitro with zidovudine, either alone or with a second nucleoside analog. It has high central nervous system excellent penetration oral bioavailability. Nevirapine induces its own metabolism, and so its dose is escalated after 14 days to its maintenance level. It can reduce indinavir levels, but this does not appear to be clinically significant. Clinical studies indicate that nevirapine is most active when used in combination with two nucleoside analogs in antiretroviral naive patients. The major toxicity is a maculopapular rash that usually occurs within the first 6 weeks of treatment and is usually mild and self-limited.
Delavirdine, the second NNRTI to be approved, is also metabolized by the cytochrome P450 3A family. Unlike nevirapine, it reduces enzyme activity in this system and therefore inhibits its own metabolism. The potential for drug interactions must therefore be kept in mind. There are not yet sufficient data regarding the safety of combining it with protease inhibitors. It has good oral bioavailability but poor central nervous system penetration and must be taken three times daily. As observed for nevirapine, rash is the most common adverse effect. Clinical trials have not shown a clinical benefit to the use of delavirdine in combination with azidothymidine or Dideoxyinosine, although improvement in surrogate markers was seen. Its ultimate role in therapy remains to be determined.
| Table Nonnucleoside Reverse Transcriptase Inhibitors | |||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||
Three other Nonnucleoside reverse transcriptase inhibitors are in various stages of development. Loviride’s clinical benefits have been small, and at this point there are no plans to market this drug. Efavirenz (DMP 266) is a very potent NNRTI with a long half-life, which allows once-daily dosing and good central nervous system penetration. It too induces the hepatic cytochrome P450 system with the potential for drug interactions. Clinical studies are under way. There are thus far insufficient data to comment on the potential for quinoxaline (HBY 097).
Other inhibitors of reverse transcriptase
Other inhibitors of reverse transcriptase include phosphonoformate (foscarnet), suramin, heteropolymer-23 (HPA-23), and rifabutin. Foscarnet is an analog of pyrophosphate. In vitro studies have shown its inhibitory activity against viral DNA polymerase from cytomegalovirus and reverse transcriptase from human immunodeficiency virus. In clinical trials, foscarnet was well tolerated and decreased p24 antigenemia. However, only an intravenous preparation is available, which makes long-term administration for human immunodeficiency virus impractical. Suramin, which has been used as an antiparasitic agent, was fortuitously found to inhibit human immunodeficiency virus reverse transcriptase in vitro. However, clinical trials could not demonstrate any clinical or virologic improvement. Additional compounds with in vitro inhibitory activity that have been clinically tested but have not been found to have clear antiviral efficacy include HPA-23, a compound containing antimony and tungsten, and the antimycobacterial agent rifabutin.
Late-stage inhibition
A number of agents interfere with late-life-cycle states of human immunodeficiency virus. These include antisense molecules, ribavirin, a-interferon, and glucosidase inhibitors. Antisense oligonucleotides are short synthetic sequences of DNA that are complementary to viral RNA sequences. They can bind to viral RNA and can inhibit translation and production of protein. These oligonucleotides can pass through cell membranes and, when modified by the addition of phosphoramidates or phosphorothioates, can resist destruction by host cellular nuclease. These oligonucleotides have also been shown to be competitive inhibitors of human immunodeficiency virus reverse transcriptase. However, a number of technical problems need to be resolved before clinical trials with these agents can be undertaken.
Ribavirin, a guanosine analog, has demonstrated some anti-human immunodeficiency virus effect in vitro, perhaps by inhibiting the guanylation step required for 5′ capping of viral RNA. Results of clinical trials with this agent have not demonstrated any antiviral efficacy.
α-Interferon, a protein produced by virally infected leukocytes, has anti-human immunodeficiency virus activity in vitro. The exact mechanism of its activity has not been elucidated; however, interferon-a may block final human immunodeficiency virus assembly and budding of mature viral particles at the cell surface. It may also have a role in inhibiting protein translation. Clinical trials of interferon-a in human immunodeficiency virus-infected patients primarily have involved treatment of Kaposi’s sarcoma. Although there is evidence that interferon-a can reduce p24 antigenemia and that synergy with azidothymidine exists in vitro, clinical trials have not shown clinical benefits above that related to the concomitant use of azidothymidine.
Before viral assembly and release, many viral proteins must be glycosylated. However, more sugars are added than are necessary, and some must be removed before release. Cellular glycosidases, primarily α-glycosidase 1, are responsible for cleaving the terminal sugars on the human immunodeficiency virus envelope glycoprotein before its release. Compounds such as castanospermine and ηbutyldeoxynojirimycin (ηbutyl DNJ) are cellular glucosidase inhibitors that have demonstrated antiviral activity in vitro. To date, no clinical trials have been published showing efficacy.
Protease inhibitors
Development of the protease inhibitors has revolutionized the treatment of human immunodeficiency virus. These very powerful agents have been associated with impressive improvements in immunological function and decreases in plasma viral load, and their use has contributed to better understanding of the pathogenesis of human immunodeficiency virus. The human immunodeficiency virus protease enzyme is critical for the posttranslational processing of the polyprotein products into core proteins and viral enzymes of the infectious virion. Inhibition of the protease results in the release of a noninfectious and immature viral particle. Four protease inhibitors are currently licensed for use. All show substantial in vitro activity against both human immunodeficiency virus-1 and human immunodeficiency virus-2 and a wide therapeutic index. They are metabolized by the hepatic cytochrome P450 (CYP) system with variable ability to induce or inhibit the enzyme. Thus, the potential for drug interactions, especially with ritonavir, must be considered. Studies have shown the importance of adequate drug levels to maintain viral suppression, suggesting that adherence to the prescribed regimen is crucial to long-term benefit. None of them should be used as monotherapy. Finally, although short-term studies have been impressive, the longer-term efficacy of these drugs is not yet known. One complication that became apparent with increased clinical use was the occurrence of spontaneous bleeding episodes in human immunodeficiency virus-infected hemophiliacs who were treated with these agents. Other important new adverse effects are the induction of glucose intolerance, redistribution of body fat giving rise to a buffalo hump and a “big belly.”
Saquinavir, the first of the protease inhibitors, has poor bioavailability and undergoes extensive hepatic metabolism. To improve its absorption, it is taken with a meal with a high fat content and with grapefruit juice (to inhibit CYP 3A). A new soft-gel formulation has been introduced as a means of improving bioavailability. It is very well tolerated, the major adverse effects being gastrointestinal and usually mild. There have been two large trials examining the use of saquinavir. In one, a combination of azidothymidine, Dideoxycytidine, and saquinavir had quite modest benefit compared with combinations of azidothymidine and Dideoxycytidine or azidothymidine and saquinavir. Although the second study documented benefit of saquinavir plus Dideoxycytidine compared with either drug alone, the comparative treatments do not represent the standard of care.
Ritonavir has the advantage of twice daily dosing, but the disadvantage of requiring storage in a refrigerator and away from direct light. Another major disadvantage is the number of potential drug interactions with ritonavir related to its effect on the cytochrome P450 system. Ritonavir has been the PI with the least tolerability. Gastrointestinal adverse effects are common even with dose escalation. Clinical studies have shown both surrogate marker and clinical benefit with ritonavir, with delayed disease progression and reduced mortality in patients with advanced disease.
Indinavir is quite well tolerated, the most important adverse reaction being nephrolithiasis. Patients should drink at least 1.5 L of fluid throughout the day to prevent this problem. It too is metabolized by and interacts with the cytochrome P450 system; so the potential for a variety of drug toxicities must be considered when patients are taking other medications in addition to indinavir. It is taken on an empty stomach for optimal absorption and should be taken regularly around the clock [Reviewing ed asks, How often?] to maintain constant therapeutic plasma concentrations. Recent trials have shown immunological and virological benefits of indinavir when used with azidothymidine and 3TC in patients with prior antiretroviral therapy and slowed progression of disease in previously treated patients with CD4 counts ≤200 cells per cubic millimeter. Its role in treating patients at earlier stages of disease has not been defined in clinical trials.
Nelfinavir is given three times daily with food. The main adverse effect is diarrhea. It inhibits cytochrome P450 (though to a lesser extent than ritonavir doses) and hence drug interactions may be problematic.
The newest protease inhibitor is GW141; it is still in clinical trials.
Antiretroviral resistance
The problem of drug resistance was noted early in the history of human immunodeficiency virus treatment and has been observed for the three classes of drugs in clinical use. Loss of antiretroviral effect with a variety of antiretroviral agents has been reported to coincide with the appearance of viral mutants with reduced drug sensitivity. There is increasing evidence that drug resistance is associated with risk of disease progression, but it is not clear whether this represents cause or effect. The occurrence of cross-resistance may impact on effectiveness of later treatments and must be considered when initiating and changing therapies. Zidovudine resistance does not appear to affect susceptibility to Dideoxyinosine, Dideoxycytidine, or 3TC, but d4T cross-resistance has been reported. Dideoxyinosine and Dideoxycytidine are cross-resistant with each other and have partial cross-resistance with 3TC. Resistance to lamivudine occurs rapidly through a single mutation conferring high-level resistance to it and partial cross-resistance with Dideoxycytidine and Dideoxyinosine. There appears to be partial cross-resistance to Dideoxycytidine, Dideoxyinosine, and azidothymidine in d4T-resistant mutants. There is no cross-resistance between nucleoside analogs and Nonnucleoside reverse transcriptase inhibitors.
High-level resistance develops rapidly to the Nonnucleoside reverse transcriptase inhibitors and is associated with considerable cross-resistance to other Nonnucleoside reverse transcriptase inhibitors. For this reason, it is recommended that the Nonnucleoside reverse transcriptase inhibitors be used only in combinations anticipated to greatly reduce the viral load. [Reviewing ed asks, Is there a principle here?]
Development of resistant mutants has been seen with saquinavir, ritonavir, indinavir, and nelfinavir and is reduced by combination with at least one nucleoside analog. Indinavir resistance is associated with saquinavir and ritonavir cross-resistance and ritonavir-resistance with indinavir and nelfinavir cross-resistance; saquinavir resistance appears to develop more slowly, and saquinavir recently has been reported cross-resistant to the other protease inhibitors. Continued study will provide more data on resistance and cross-resistance patterns and how to sequence therapies to avoid and anticipate problems with cross-resistance.
Guidelines for the use of antiviral agents in human immunodeficiency virus infection
The treatment of human immunodeficiency virus infection has changed dramatically over the last decade. These changes have come about as a result of a better understanding of the pathogenesis of infection and of the pharmacokinetics and resistance patterns of the available drugs. No doubt management will continue to evolve as new information becomes available.
The new combination therapies look very promising in early trials. Theoretically, combination therapy for human immunodeficiency virus could exert enhanced activity by affecting different stages of viral replication. Also, if the drugs had different toxicities, it might be possible to use lower doses of each individual compound while maintaining antiviral activity. Synergism has been noted, in vitro and in vivo, for a number of different combinations of antiretrovirals. Additionally, several clinical trials have demonstrated improved clinical and/or surrogate marker outcomes of combination therapy as compared with monotherapy. Combinations of antiretroviral therapy have thus become standard for anti-human immunodeficiency virus treatment. Commonly used combinations to treat human immunodeficiency virus infections.
| Table Characteristics of Protease Inhibitors | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Table Drugs That Should Not Be Used with Protease Inhibitors | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Principles
Key issues to be considered in relation to anti-human immunodeficiency virus treatment are
- Even the clinically latent phase of human immunodeficiency virus infection is marked by intense viral replication.
- The relation between prognosis and viral load has been demonstrated.
- Beneficial effects on immunological and virologic markers are seen with therapy.
- The development of resistance has been documented for all classes of agents in use.
- Cross-resistance within groups is common.
Therapy is recommended for individuals symptomatic with human immunodeficiency virus with CD4+ T-cell counts <500 cells/mm3 or with a viral load ≥10,000 human immunodeficiency virus RNA copies/mL.
Changes in therapy may be required primarily for three reasons: treatment failure, drug intolerance, or nonadherence. Treatment failure is generally recognized by return to pretreatment levels of viral load and CD and clinical progression. Appearance of any degree of viremia, however, may be a marker of treatment failure if it had previously been undetectable. Treatment failure usually poses more of a therapeutic challenge than does toxicity. Factors to be considered in choosing an alternate regimen are: prior treatments (with cross-resistance implications),
currently available drugs, previous toxicities, stage of disease, concomitant medication (potential for drug interactions), and reason for making the change (toxicity, adherence or failure). In patients who experience treatment failure, generally the most potent available regime is recommended with a change in at least two, and preferably three, drugs. The use of more intensive antiretroviral therapies has resulted in a decline in human immunodeficiency virus-related mortality from 29.4 per 100 person-years in 1995 to 8.8 per 100 person-years in the second quarter of 1997. [Au: more recent data available?]
| Table Drug Interactions between Protease Inhibitors and Other Drugs; Drug Interactions Requiring Dose Modifications | |||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||
The one clinical trial evaluating antiretroviral therapy of primary human immunodeficiency virus showed that zidovudine slowed clinical progression. Currently, it is recommended that combination therapy be used in treating primary human immunodeficiency virus: at least 2 nucleoside analogs with the addition of a PI or NNRTI considered.
Treatment of human immunodeficiency virus infection in pregnancy
Just over 2 million of the 5.2 million (40%) adults newly infected with human immunodeficiency virus in 1997 were women, most of whom were of childbearing age. It is estimated that 1. 1 million children worldwide are living with acquired immunodeficiency syndrome, 90% of whom have acquired the infection through vertical transmission (CDC 1995b).
Vertical transmission of human immunodeficiency virus can occur in utero, intrapartum, or through postpartum breast feeding. Although human immunodeficiency virus transmission can occur to the fetus as early as the eighth week of gestation, 50 to 70% of vertical transmission occurs just before or during parturition.
Use of azidothymidine during pregnancy can reduce transmission from mother to child. The acquired immunodeficiency syndrome Clinical Trials Protocol 076 was a randomized, multicenter double-blind, placebo-controlled trial in which human immunodeficiency virus-infected azidothymidine-naive pregnant women, between 14 and 34 weeks of gestation, with CD4 counts ≥200 cells/mm3 were randomly assigned to receive placebo or azidothymidine 100 mg orally 5 times per day from enrollment until labor. Azidothymidine was given intravenously during labor in a dose of 1 mg/kg per hour (a 2 mg/kg loading dose was administered over 1 hour) until delivery, The baby was given 2 mg/kg of azidothymidine syrup every 6 hours for 6 weeks beginning 8 to 12 hours after birth. In this study the rate of vertical transmission of human immunodeficiency virus was reduced from 25.5% in the placebo group to 8.3% in the azidothymidine group. Follow-up to 5 years of age showed no toxicity in the treated infants, and the rate of congenital anomalies was equal in the treated and placebo groups. The efficacy of azidothymidine in reducing vertical transmission has been shown regardless of maternal CD4 count, duration of rupture of membranes, mode of delivery or gestational age.
| Table Drug Interactions: Protease Inhibitors and Nonnucleoside Reverse Transcriptase Inhibitors — Effect of Drug on Levels and Dose | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Table Recommended Antiretroviral Agents for Treatment of Established human immunodeficiency virus Infection | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Currently combination therapy with antiretrovirals that have good placental passage (azidothymidine, 3TC, d4T, nevirapine) is recommended. There are a number of trials planned or ongoing to evaluate the safety and efficacy of combination regimens under a variety of circumstances and in a broader spectrum of pregnant patients. Some questions to be answered are whether shorter regimens may be equally effective and what treatments are best for women who have received prior antiretroviral treatment.
Postexposure prophylaxis
Occupational injuries involving percutaneous exposure to an human immunodeficiency virus-infected source have a 0.3% risk of human immunodeficiency virus transmission. A retrospective study found that increased risk of transmission was positively related to deep injury, visible blood on sharp device, device used to enter a blood vessel, and source patient having terminal acquired immunodeficiency syndrome. Risk of infection was reduced by the prophylactic use of zidovudine (CDC 1995). Current guidelines for occupational prophylaxis of human immunodeficiency virus include a combination of two or three drugs, depending on the type of exposure and source. It is recommended that treatment be started within hours, and the standard duration is 4 weeks (CDC 1996). Each institution should develop its own postexposure prophylaxis protocol based on the current recommendations.
| Table Guidelines for Changing an Antiretroviral Regimen for Suspected Drug Failure | ||||||||||||||||||||
|
||||||||||||||||||||
| Table Possible Regimens for Patients Who Have Failed Antiretroviral Therapy: A Work in Progress.a | ||||||||||||||||||||
|
||||||||||||||||||||
| Table Preclinical and Clinical Data Relevant to Use of Antiretrovirals during Pregnancy | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
No recommendations have been published on the use of postexposure prophylaxis in nonoccupational situations (e.g., after sexual and drug-use exposures). However, some physicians are recommending postexposure prophylaxis after unprotected receptive and insertive anal and vaginal intercourse with a partner who is or is likely to be human immunodeficiency virus infected. Prophylaxis is also offered for receptive fellatio with ejaculation. Currently treating only isolated exposures or exposures in a patient who is willing to practice safer behaviors in the future is suggested. A recent report from CDC (CDC 1998) defines principles of therapy of human immunodeficiency virus infection and gives guidelines for the use of antiretroviral agents in human immunodeficiency virus-infected adults and adolescents.
Immunomodulation
Human immunodeficiency virus infection is associated with many alterations in immune function. These include the well-recognized decrease in CD4+ T cells as well as altered CD4+ lymphocyte function. There is an initial increase in CD8+ lymphocyte numbers and CD8+ cell activation. Changes are also seen in lymphocyte proliferation, IL-2 and IFN-γ production, and macrophage and dendritic cell function. Thus, immune reconstitution is an important component of anti-human immunodeficiency virus management.
In addition to specific agents targeted against human immunodeficiency virus, attempts have been made to either restore or augment the host’s immune function. IL-2 is a glycoprotein product of T cells. Production of this cytokine results in proliferation of circulating T cells and activation of natural killer cells. IL-2 has been shown to produce increases in CD4+ T-cell counts. Other limitations of IL-2 are the need for intravenous or subcutaneous infusions and considerable adverse effects. Its role in treatment regimens has yet to be determined.
Administration of intravenous immunoglobulin has resulted in some reduction in bacterial infections in human immunodeficiency virus-infected children (most of whom were also receiving prophylactic antibiotics) but no improvement in mortality. No clear-cut benefit has been shown for use of intravenous immunoglobulin in human immunodeficiency virus-infected adults . However, increases are short-lived when treatment is stopped.
Isoprinosine, an immune enhancer, has some mild intrinsic antiviral activity. However, in vitro data have shown that it increases T-cell proliferation and IL-2 production. An earlier clinical trial demonstrated delayed progression to acquired immunodeficiency syndrome in a select patient population, but it has not been studied further and is not licensed for use as a treatment for human immunodeficiency virus infection.
Pentoxifylline, a tumor necrosis factor-α inhibitor, has not been shown to have an effect on CD4 counts. Studies are under way to evaluate the impact of thalidomide as an immunomodulator.
Bone marrow transplantation was reported to be effective in one patient who had received concomitant azidothymidine. However, the patient developed a malignancy and died a short time after the transplant. Furthermore, the results of bone marrow transplantation on a small series indicate that it is not effective for curing acquired immunodeficiency syndrome.
Vaccine strategies
For many viruses, administration of a vaccine has resulted in effective active immunity in the host against the specific viral agent. Given the extent of the human immunodeficiency virus epidemic and, for most infected persons, the prohibitive cost of therapy, prevention and eradication through immunization represents the ultimate goal. To date, this has not been easily achievable. human immunodeficiency virus has presented complicated problems in vaccine development including the following: the genetic variability of the virus, incomplete knowledge concerning the immunological correlates of protection, phenotypic differences between viruses that may be associated with different immune responses and mechanisms of transmission, and inability to extrapolate results of animal vaccine experiments to humans.
Most of the early efforts at developing a vaccine involved preparations of envelope gp120 or gp160 subunits, either recombinant, native, or synthetic proteins. These vaccines induced neutralizing antibodies but no cytotoxic T-lymphocyte response. In addition, it was found that these antibodies did not work as well against primary human isolates as they did against laboratory isolates, which obviously do not represent the major circulating strains. On the other hand, later vaccine experiments of recombinant live vectors expressing the human immunodeficiency virus-1 envelope gp160 induced good CTL responses but poor neutralizing-antibody response. There is good evidence that cytotoxic T lymphocytes have a major role to play in control of human immunodeficiency virus infection, and it is likely that both humoral and cell-mediated immune responses play a significant role in protection and control of early infection. Vaccine development will have to build on this concept. The 1990s saw loss of interest in vaccine approaches. However, improved understanding of human immunodeficiency virus pathogenesis has opened up new areas of experimentation, which will likely lead to testing of new candidate vaccines within the next decade.
Prevention of Opportunistic Infections in Persons Infected with Human Immunodeficiency Virus
Tables A-1 and A-2 give the current recommendations for prevention of opportunistic infections in those who are infected with human immunodeficiency virus.
New information on management of human immunodeficiency virus infection and its complications
Since new data are constantly emerging on the management of human immunodeficiency virus infection, this chapter can only serve as a basis for building an understanding of the principles of therapy of human immunodeficiency virus infection. The most up-to-date information should be obtained from the acquired immunodeficiency syndrome Treatment Information Service at http://www.hivatis.org.
Management of the Febrile Neutropenic Patient
Management of Bacterial Meningitis
Management of Infective Endocarditis
Management of Tuberculosis
Management of Malaria
Erythrocytic asexual phase
Evaluation of the efficacy of vaccines
Penicillins
β-Lactamase inhibitors
In an effort to avoid bacterial mechanisms of resistance to penicillins, there has been a considerable effort to develop substances that inhibit β-lactamase. Clavulanic acid, tazobactam, and sulbactam are commercially available β-lactamase inhibitors. They bind to conserved regions within the β-lactamase produced by a variety of organisms. The binding alters the structure of the enzyme, thereby preventing it from binding and hydrolyzing the β-lactam of the antibiotic. They have been effectively used in the treatment of non-life-threatening mixed infections (e.g., aspiration pneumonia, diabetic foot ulcers, and intra-abdominal and pelvic sepsis). Clavulanic acid is combined with preparations of ticarcillin and amoxicillin, tazobactam with piperacillin, and sulbactam with parenteral ampicillin. These preparations improve the spectrum of activity against most anaerobes, staphylococci, and certain strains of aerobic gram-negative bacteria.
Cephalosporins
Cephalosporins kill bacteria by interfering with synthesis of their cell walls. They are most commonly used in hospitalized patients for prophylaxis against surgical wound infections because of their broad spectrum of activity. The cephalosporins are divided into groups based on their spectra of activity.
First-generation cephalosporins are effective against susceptible aerobic gram-positive staphylococci, gram-negative bacteria, and streptococci. They are useful in most cases of surgical prophylaxis and in minor to moderate skin, respiratory, and urinary tract aerobic gram-positive and gram-negative bacterial infections. First-generation cephalosporins have no place in the treatment of mixed infections because they are ineffective against anaerobes.
Second-generation cephalosporins have less activity against aerobic gram-positive bacteria but enhanced aerobic gram-negative and anaerobic bacterial coverage compared with first-generation cephalosporins. They are most appropriately used to treat mixed infections, including intra-abdominal and pelvic sepsis, diabetic foot ulcers, aspiration pneumonia, many abscesses in different anatomic sites, and other polymicrobial infections. In the treatment of mixed anaerobic-aerobic infections, the most common error is the failure to consider surgical intervention (e.g., débridement of dead tissues or a surgical procedure to drain an abscess).
The third-generation cephalosporins are more effective against aerobic gram-negative organisms than their precursors. However, they are unreliable against aerobic gram-positive and most anaerobic bacteria. Because of their unique pharmacokinetic properties, third-generation cephalosporins are most useful to treat aerobic gram-negative bacterial meningitis and biliary tract infections. They should not be used as monotherapy to treat mixed infections or as empirical therapy for serious bacterial infections when staphylococci, streptococci, or anaerobes might be the etiologic agents. The overutilization of all cephalosporins has resulted in increased rates of enterococcal superinfections, because these microorganisms are not eradicated by this entire class of antibiotics. Within this class certain agents have enhanced activity against a particular organism or class of organisms. For example, cefotaxime has more activity against gram-positive cocci (except enterococci, of course); ceftizoxime against anaerobes; and ceftazidime is most active against Pseudomonas aeruginosa.
Monobactams
Aztreonam is the only currently approved monobactam. It kills susceptible microorganisms by binding to penicillin-binding proteins of the bacteria, ultimately interfering with cell-wall synthesis. The spectrum of activity of aztreonam is limited, exhibiting activity against only aerobic gram-negative bacteria causing septicemia, pneumonia, osteomyelitis, and urinary tract infections. Aztreonam is active against enterobacteriaceae, Yersinia spp., Pasteurella multocida, Capnocytophaga spp., Plesiomonas spp., Aeromonas spp., H. influenzae and Neisseria spp. The MIC90 against P. aeruginosa is 16 mg/L, a value high enough to result in only minimal activity against this microorganism. This agent has little activity against aerobic gram-positive and anaerobic bacteria. Therefore, this drug cannot be used in the majority of infectious syndromes. This relatively safe but expensive agent should be used in patients with aerobic gram-negative bacterial infections who have renal insufficiency and require prolonged therapy. The value of this regimen is that aminoglycoside therapy, which could further compromise renal function, will not have to be initiated in such compromised patients. Another benefit of aztreonam therapy is that it can be administered safely in patients who have had previous anaphylactic reactions to penicillin.
Carbapenems
Carbapenems are a group of β-lactams with a carbapenem nucleus. Imipenem and meropenem are the only available drugs in this class. They bind to all the penicillin-binding proteins but preferentially bind to PBP2 and PBP1, which are responsible for maintaining the bacterium’s constant diameter and extending its cell wall in any direction respectively. Interference with these bacterial transpeptidases leads to rapid lysis of most anaerobic and aerobic bacteria.
Imipenem has the widest spectrum of activity of the currently available β-lactams, and its usage ordinarily should be reserved for multidrug-resistant bacteria. Imipenem appears to be effective only against extracellular bacteria; it should not be used to treat intracellular pathogens (e.g., L. monocytogenes). Because of imipenem’s broad spectrum of antibacterial activity, it probably is worth remembering that the only bacterial isolates that are not susceptible to imipenem include methicillin-resistant staphylococci, Enterococcus faecalis, Stenotrophomonas maltophilia, B. cepacia, and rare groups of Bacteroides (e.g., B. ovatus, B. disiens, and B. thetaiotaomicron). Imipenem is not recommended as routine monotherapy to treat bacterial infections of the lower respiratory tract, osteomyelitis, bacterial septicemia and endocarditis, or urinary tract, skin, or intra-abdominal infections.
Carbapenems are extensively metabolized by the renal tubular brush border dipeptidase, dehydropeptidase-1. A selective competitive antagonist of this enzyme has been identified (cilastatin), and when it is combined with imipenem in a 1:1 ratio, the antibiotic persists in the plasma for prolonged periods. Imipenem rarely produces toxic effects. High doses can produce convulsions in patients with renal insufficiency who have a prior history of seizure disorders. Imipenem can be made safe even in these patients if the rate of administration is slowed and
the dose is reduced in proportion to the extent of renal insufficiency.
Meropenem has a spectrum of antimicrobial activity that is similar to imipenem. However, it is somewhat less active against gram-positive cocci and more active against Enterobacteriaceae and P. aeruginosa than imipenem. Meropenem has relatively greater stability against human dehydropeptidase-1; so concomitant administration of cilastatin is not necessary. It has less potential for inducing seizures compared with imipenem.
Aminoglycosides
Vancomycin/Glycopeptides
Chloramphenicol
Chloramphenicol exerts its broad antibacterial effects by binding to the 50-S ribosome subunit, inhibiting protein synthesis. Because of its serious toxic profile, which includes aplastic anemia and gray baby syndrome, and the availability of other less toxic but equally effective drugs used for similar indications, chloramphenicol is not extensively used.
Tetracyclines
The molecular structure of tetracyclines includes four benzene rings. They have a broad spectrum of antibacterial activity, and by binding to the 30-S ribosome subunit, they exert their effects by inhibiting protein synthesis. A considerable number of compounds, especially orally active preparations, have been developed. Unfortunately, because of the high prevalence of tetracycline-resistant microorganisms and the availability of alternative effective antibiotics, the place of tetracyclines in therapy has diminished. Tetracyclines remain the drugs of choice to treat brucellosis, which also requires combination with streptomycin. They also are the drugs of choice to treat chlamydial and rickettsial infections and melioidosis. Doxycycline is effective against some strains of Mycobacterium fortuitum-chelonei. Since resistance to tetracyclines is so prevalent among all species of bacteria, the effectiveness of tetracyclines in treating a variety of clinical syndromes cannot be predicted.
Major tetracycline toxicity includes nephrotoxicity and hepatotoxicity, especially in the pregnant patient. Tetracyclines are contraindicated in pregnant women and in children <8 years of age because of accumulation of these drugs in the growth zone of teeth (also in bones), producing a characteristic unpleasant pigmentation.
Macrolides
Clindamycin
Clindamycin is a chemically modified derivative of lincomycin. It exerts its activity by binding to the 50-S subunit of the bacterial ribosome, inhibiting protein synthesis. Its antibacterial spectrum includes staphylococci and streptococci (but not enterococci), many anaerobic gram-positive strains, and most anaerobic gram-negative bacteria. Clindamycin is most appropriately used to treat clinical syndromes that involve anaerobic pathogens. Clindamycin is not recommended as primary agent to treat staphylococcal or streptococcal infections despite its antibacterial activity against these microorganisms.
Clindamycin is extensively cleared by the liver. Doses should be modified when patients have significant hepatic insufficiency. The most common serious adverse reaction to clindamycin is pseudomembranous colitis. This effect occurs 2 days to 3 weeks after beginning therapy in 1% to 10% of patients treated with clindamycin. However, as was discussed earlier, this example of superinfection is not unique to the use of clindamycin, since essentially any antibiotic can produce the same condition. The pathogenesis of this syndrome generally involves excretion of the antibiotic in the stool and selection for C. difficile in the gastrointestinal tract. Clinically important C. difficile strains elaborate toxins that are cytotoxic to the epithelial mucosal cells, and a pseudomembrane covers the afflicted area. Symptoms of colitis usually include fever, cramping, abdominal pain, and diarrhea. Appropriate diagnostic tests for this syndrome include sigmoidoscopy or colonoscopy (in some situations), assessment of the stool for the presence of inflammatory cells, and detection of the toxin in the stool. Effective therapy for this superinfection includes oral metronidazole or oral vancomycin in resistant cases. Cholestyramine can also be used to bind the toxin. Caution must be taken to ensure that cholestyramine does not bind other medications that the patient is receiving — hence it should be administered 2 hours before or after other medications.
Metronidazole
Metronidazole is a nitroimidazole drug that has activity against anaerobic bacteria and protozoan. The drug is postulated to be metabolized within the anaerobe to an active drug that interacts with the anaerobe’s DNA to produce cell death. The spectrum of antibacterial activity for metronidazole is confined to most anaerobes. It has essentially no activity against aerobic bacteria. Metronidazole has been effectively used to treat a variety of infections involving anaerobic strains, including intra-abdominal sepsis, genital infections, abscesses, aspiration pneumonia, and osteomyelitis. It has also been used to treat trichomoniasis, amoebiasis, giardiasis, and bacterial vaginosis. Because of its low resistance rate and low cost, metronidazole should be the drug of choice in treatment of many anaerobic infections.
Sulfonamides and Trimethoprim
Sulfonamides and trimethoprim are used to treat mild to moderately severe bacterial infections caused by sensitive organisms. They exert their antibacterial effects by interfering with the microorganism’s folate metabolism, which is essential for purine and ultimately DNA synthesis. There are many sulfonamides available, differing from each other by their duration of action. Sulfadiazine is short-acting (must be administered every 6 to 8 hours), whereas sulfamethoxazole has medium duration of action (given every 12 hours), and sulfadoxine is ultralong-acting (requires dosage once a week). Most sulfonamides are readily absorbed from the gastrointestinal tract.
Trimethoprim is an inhibitor of dihydrofolate reductase and frequently is combined with sulfonamides, but it can also be used as a single agent with success equivalent to that expected of sulfonamides used alone. The two agents once had a wide range of antimicrobial activity. However, extensive usage and the rapid development of microbial resistance to these drugs have narrowed their spectrum of activity.
Sulfonamide-susceptible microorganisms include some staphylococci, many streptococci except S. faecalis, many anaerobic gram-positive bacilli, some L. monocytogenes, most species of Nocardia, the majority of Enterobacteriaceae, many pathogenic species of Neisseria, Stenotrophomonas maltophilia, H. influenzae, many other gram-negative anaerobes, strains of Chlamydia except C. psittaci, some atypical species of mycobacteria, protozoa such as Toxoplasma gondii and malaria, and unique fungi such as P. carinii when therapy combines pyrimethamine or trimethoprim with sulfonamides.
Trimethoprim-sulfamethoxazole is an effective combination agent for prophylaxis against P. carinii infection in human immunodeficiency virus patients when the CD4 count is <200/mm3. In such instances one double-strength tablet is administered once daily or three times weekly.
The combination trimethoprim-sulfamethoxazole is effective in treating urinary tract infections that are caused by a variety of aerobic gram-positive and gram-negative bacteria, otitis media, acute and chronic bronchitis, and bacterial pneumonia, venereal diseases (e.g., gonorrhea, chancroid, granuloma venereum, and sometimes C. trachomatis infections), typhoid fever from susceptible strains, shigellosis, cholera, brucellosis, nocardiosis, toxoplasmosis, and P. carinii pneumonia. Allergic adverse responses are frequent, especially in acquired immunodeficiency syndrome patients. They most notably include rashes, drug fever, and bone marrow suppression.
Quinolones
Norfloxacin and ciprofloxacin were the first of the quinolones approved by the FDA. They are derivatives of nalidixic acid, which is the prototype compound of this class. In contrast to nalidixic acid, norfloxacin and ciprofloxacin readily penetrate the outer membranes of a large number of gram-negative and selected gram-positive bacteria. These agents exert their antibacterial effects by binding to DNA gyrase, thereby inhibiting replication of bacterial DNA. Microorganisms are killed by quinolones if they continue to synthesize protein. Ultimately, the bacteria cannot divide because of the effects of the quinolone. Resistance develops via chromosomal mutation, especially among enteric flora exposed to subinhibitory concentrations of drug.
These drugs are administered orally. Norfloxacin, which is poorly absorbed, is more limited in its distribution within the body than is ciprofloxacin. Norfloxacin is used mainly to treat urinary tract infections. Ciprofloxacin, in addition to being useful in treating urinary tract infections, is used to treat bone, respiratory, inner ear, and soft tissue infections because of its spectrum of activity and excellent oral bioavailability.
The quinolones exert their greatest antibacterial effects against susceptible aerobic gram-negative bacteria from the Enterobacteriaceae, including many strains of P. aeruginosa. Infections due to these microorganisms are appropriately treated with quinolones. The role of quinolones in the treatment of other gram-negative microorganisms (e.g., H. influenzae and N. gonorrhoeae) is controversial. Quinolones are not recommended to treat anaerobic infections, nor as monotherapy for serious life-threatening infections or meningitis.
A number of new quinolones so-called “respiratory quinolones” because of enhanced activity against S. pneumoniae compared with ciprofloxacin, are now available. These include levofloxacin, sparfloxacin, grepafloxacin, and trovafloxacin. Since these agents are active against penicillin- and/or macrolide-resistant S. pneumoniae, there will be a tendency to use these newer quinolones widely in the treatment of community-acquired pneumonia. At present we have very little data with which to make clear recommendations about the superiority of one class of antibiotics over another for the ambulatory management of community-acquired pneumonia. Grepafloxacin has been removed from the market because of torsade de pointes due to prolonged QT interval. Trovafloxacin has been removed because of severe hypersensitivity hepatitis, some cases of which were fatal.
Streptogramins
This is a distinct class of antibiotics consisting of two structurally unrelated compounds that interact synergistically. Group A streptogramin is a polyunsaturated macrolactone with a molecular mass of 500 daltons, whereas group B streptogramin is a cyclic hexadecapeptide with a molecular weight of 800 daltons. Streptogramins work at the ribosome level. Group A streptogramins inhibit both the binding of peptidyl-tRNA to the donor site and that of aminoacyl-tRNA to the acceptor site of peptidyltransferase. Group B streptogramins prevent peptide bond formation indirectly, causing the release of peptidyl-tRNA from the donor site. Currently there is one antibiotic, quinapristin/dalfopristin (Synercid), of this class available in Canada (not yet approved by the FDA). This combination consists of 30% quinapristin and 70% dalfopristin. Streptogramins typically are active against both methicillin-susceptible and methicillin-resistant S. aureus and S. epidermidis, penicillin-susceptible and penicillin-resistant S. pneumoniae, E. faecium both vancomycin-sensitive and resistant strains, group A, B, C streptococci, Corynebacterium jeikeium, Mycoplasma species, Legionella spp, H. influenzae, M. catarrhalis, Neisseria spp. Quinapristin/dalfopristin is available for intravenous use only. It is given as 7.5 mg/kg every 8 hours or every 12 hours. Nausea, vomiting, and skin rash are encountered with a frequency similar to other antibiotics. Phlebitis at the site of the infusion is commonly encountered and leads to discontinuation of treatment in about 10% of patients. Asymptomatic hyperbilirubinemia is seen in 10%. Quinapristin/dalfopristin is metabolized via the cytochrome P450 system and hence can interact with drugs such as FK506 that are metabolized via the same pathways when given concurrently. Currently quinapristin/dalfopristin is reserved for resistant gram-positive infections, especially vancomycin resistant E. faecium.
| Table Comparison of Selected Features of “Respiratory Quinolones” with Ciprofloxacin | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Oxazolidinones
Linezolid is the first synthetic oxazolidinone antibiotic. It binds to the 505 ribosomal subunit. It is active against vancomycin resistant enterococci, and against other streptococci as well as S. aureus and coagulase negative staphylococci.
Antituberculosis Drugs
Treatment of mycobacterial infections requires multiple drugs because monotherapy frequently fails. Resistance of M. tuberculosis to an antimicrobial agent occurs as a result of spontaneous mutation, at a usual frequency of 1 in 105 to 106. Mutational resistance to each drug occurs as an independent event, and thus the likelihood of resistancy by a single organism being to two drugs is equal to the product of the individual probabilities. Therefore, combined chemotherapy with two or more drugs prevents the emergence of strains resistant to an individual drug. The likelihood of organisms developing resistance is increased if the patient previously has been treated and if the infection was the result of exposure to a resistant stain.
Tuberculocidal drugs are preferred and should be capable of killing both rapidly dividing extracellular organisms and slower dividing intracellular organisms in order to prevent relapse. Isoniazid and rifampin are tuberculocidal in both intra- and extracellular locations and are generally recommended in all patients with mycobacterial infection. Streptomycin is tuberculocidal for extracellular organisms only, whereas pyrazinamide is tuberculocidal for intracellular organisms. Ethambutol, p-aminosalicylic acid, and ethionamide are only tuberculostatic.
The institution of effective chemotherapy results in rapid reversal of infectiousness of mycobacteria within 3 to 7 days. However, the clinical manifestations of disease may persist for prolonged periods. Although antimicrobial-resistant mycobacterial strains emerge as an important epidemiologic cause for clinical failure, the major cause of failure of an effective drug regimen for mycobacterial infections is lack of compliance by the patient.
Antifungal Drugs
Antiviral Agents
In the last decade, considerable strides have been made developing effective antiviral therapy. A major obstacle in such development has been identifying agents that do not injure host cells but still effectively inhibit viral metabolism and replication. The clinically most important new antiviral agents include famciclovir, valacyclovir, foscarnet, and cidofovir for treating herpes viruses; azidothymidine and a variety of new antiretroviral drugs for the human immunodeficiency virus agent (see the section on treatment of acquired immunodeficiency syndrome); and ganciclovir to treat infections caused by cytomegalovirus.
Acyclovir is a purine nucleoside analog. Its antiviral activity is almost completely restricted to the herpesviruses (HSV 1 and 2, VZV, and Epstein-Barr virus). Once acyclovir has penetrated virally infected cells, it is phosphorylated into acyclovir monophosphate by HSV thymidine kinase and sequentially into di- and triphosphate forms by cellular enzymes. Acyclovir is a potent inhibitor of viral DNA polymerases and also terminates biosynthesis of the strand of viral DNA. Acyclovir distributes widely throughout the body including the cerebrospinal fluid. The major route of elimination is via glomerular filtration and tubular secretion. Therefore, dosage adjustment is required in patients with renal dysfunction. Acyclovir is effective in the treatment of a number of infections caused by herpes simplex virus, including mucocutaneous, genital, and encephalitic infections, and varicella-zoster infections.
Valacyclovir, the L-valyl ester of acyclovir, is almost entirely converted to acyclovir after oral administration. Valacyclovir has bioavailability 3 to 5 times as great as that of acyclovir and is given at a dose of 1 gram three times daily. Some immunocompromised patients have developed thrombocytopenic purpura/hemolytic-uremic syndrome while receiving high-dose valacyclovir.
Ganciclovir (DHPG) is also a nucleoside analog. Its antiviral activity is primarily directed against cytomegalovirus. Once phosphorylated to the triphosphate form by cellular enzymes, it interferes with viral replication through competitive inhibition of viral DNA polymerase and also terminates viral DNA synthesis. Ganciclovir is well distributed to most organs including lungs, liver, and the brain and is primarily excreted by the kidneys. Dosage adjustment is also indicated in patients with renal dysfunction. The primary indication of ganciclovir is the treatment of cytomegalovirus-associated retinitis in the immunocompromised host. Data on ganciclovir treatment of cytomegalovirus-associated pneumonitis, encephalitis, colitis, and hepatitis remains controversial.
Famciclovir is the diacetyl, 6-deoxyester of the guanosine analog penciclovir. It is well absorbed (bioavailability 77%) and rapidly converted to penciclovir. It is administered every 8 hours with adjustment for renal insufficiency. It is approved for the treatment of herpes zoster and recurrent genital HSV infections.
Foscarnet (phosphonoformic acid) is a pyrophosphate-containing compound with activity against herpes viruses including cytomegalovirus. It can only be administered intravenously and is infused over 1 to 2 hours using an infusion pump. Administration of 500 mL of saline before foscarnet lessens renal toxicity. The major indications for foscarnet are cytomegalovirus retinitis, and HSV infections resistant to acyclovir. In a comparative trial, foscarnet was as efficacious as ganciclovir for the treatment of cytomegalovirus retinitis but was associated with longer survival (Ocular Complication of acquired immunodeficiency syndrome Research Group 1992). Renal function should be monitored carefully during treatment with foscarnet because of its nephrotoxicity. It binds divalent cations, and hence hypocalcemia, hypomagnesemia, hypokalemia, and hypo-or hyperphosphatemia can develop. Uncircumcised males can develop balanitis due to irritant action of foscarnet excreted in the urine. Careful drying of the penis following urination can prevent foscarnet-induced balanitis.
Cidofovir, a phosphonomethylether derivative of cytosine, is highly active against cytomegalovirus, including some ganciclovir-resistant and foscarnet-resistant strains. It is mostly cleared by the kidney and has a half-life of 2.6 hours. It is given intravenously (ocular implants are also available). Concomitant administration of probenecid markedly prolongs the half-life of cidofovir and protects against nephrotoxicity. The intracellular half-life of cidofovir diphosphate is 17 to 30 hours, and hence, it can be administered once a week to once every two weeks.
The new antiretrovirals are described in the section on the treatment of acquired immunodeficiency syndrome.
The use of interferons as antiviral agents has been mainly for treatment of hepatitis B and C, although IFN-a has also been used for the treatment of Kaposi’s sarcoma and hairy-cell leukemia. The administration of interferon alpha 2b (5 million units daily for 16 weeks) to patients with chronic hepatitis B infection resulted in loss of markers of HBV replication in 33- to 37% of cases, whereas 10- to 20% became hepatitis B surface antigen negative. Several interferon preparations (alpha 2a, alpha 2b and αL [lymphoblastoid]) have been studied for the treatment of hepatitis C.

