Nucleoside analog reverse transcriptase inhibitors have formed the backbone of anti-human immunodeficiency virus therapy for the last decade (see Table Characteristics of Nucleoside Reverse Transcriptase Inhibitors). Reverse transcription is necessary for human immunodeficiency virus RNA to be used as a template to produce viral DNA, which can be integrated into the cellular genome. In order for reverse transcription to take place, deoxynucleotide must be added to the end of the elongating DNA. A 3′-hydroxyl group is present on the sugar moiety of deoxynucleotides that can form a 3′,5′-phosphodiester bond allowing further addition of nucleotide triphosphates.
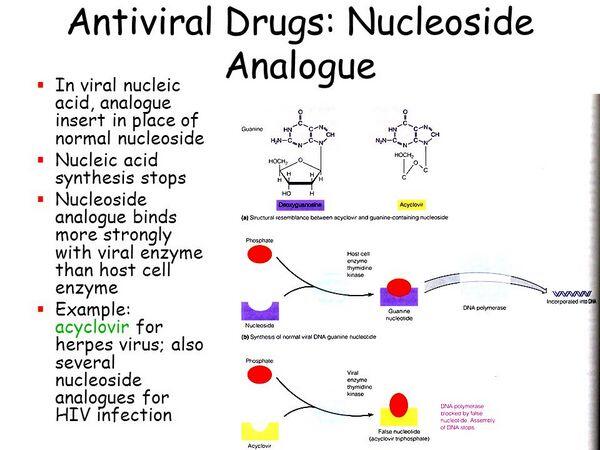
Several deoxynucleotide analogs have been synthesized that differ from normal deoxynucleotides in that the 3′-hydroxyl group of the sugar moiety (deoxyribose) has been replaced with another molecule (e.g., an azido group in the case of azidothymidine). These substitutions do not allow addition of further nucleotides in the process of chain elongation. In order for nucleoside analogs to exert their effect, they must be converted to their active triphosphate nucleotides by cellular kinases.
Zidovudine (azidothymidine), a 3′-azidothymidine derivative, was the first drug to receive FDA approval for the treatment of human immunodeficiency virus. Originally synthesized in the early 1960s, it was found to have in vitro anti-human immunodeficiency virus activity in 1985. It has good central nervous system penetration. A number of studies have evaluated the efficacy of azidothymidine. In brief, they demonstrate
- prolonged survival in patients with acquired immunodeficiency syndrome and acquired immunodeficiency syndrome-related complex (ARC),
- equal efficacy of a large dose (1200 to 1500 mg/day) versus half that dose (500 to 600 mg/day),
- decreased toxicity in patients receiving half-dose azidothymidine, and 4) clinical improvement in children with symptomatic human immunodeficiency virus infection.
The benefit of azidothymidine in the treatment of patients with CD4 counts between 0.20 and 0.50 × 109/L was less clear with some studies demonstrating slowed progression to acquired immunodeficiency syndrome, whereas other studies showed no long-term progression or survival benefit. Trials have not shown a treatment benefit in individuals with CD4 counts >0.5 × 109/L (Volberding et al. 1990; Concorde Coordinating Committee 1994).
Studies have since clearly demonstrated better outcomes with combination therapies including zidovudine as compared to zidovudine monotherapy. The role for zidovudine is currently as part of a combination regime. azidothymidine alone reduces the rate of vertical transmission of human immunodeficiency virus (from mother to child) during pregnancy. The major adverse reaction associated with azidothymidine administration is bone marrow toxicity that usually appears 4 to 12 weeks after initiating therapy and is more common in patients taking higher doses and with more advanced disease.
| Table Characteristics of Nucleoside Reverse Transcriptase Inhibitors | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Dideoxycytidine, a pyrimidine analog also known as zalcitabine, is the most potent of this group of drugs in vitro. Early data showed that patients who had not previously received antiretroviral therapy did more poorly on initial Dideoxycytidine than on initial zidovudine. Most trials, however, have focused on the use of Dideoxycytidine in combination therapy, where benefit has been shown as compared to zidovudine monotherapy in antiretroviral naive patients. The major toxicity of Dideoxycytidine is a painful peripheral neuropathy that is dose dependent and cumulative. It is generally slowly reversible once the drug is withdrawn. Other toxicities include oral aphthous ulcers and pancreatitis.
Dideoxyinosine or didanosine is a purine analog. The acid instability of Dideoxyinosine requires that the oral form be buffered for optimal intestinal absorption; Dideoxyinosine was initially approved for use in patients with advanced human immunodeficiency virus who were intolerant of or resistant to azidothymidine. As with Dideoxycytidine, its greatest potential seems to be as part of a combination regime.
The drug has also been shown to have benefit in children, either alone or in combination with zidovudine. The major dose-limiting toxicity with Dideoxyinosine appears to be peripheral neuropathy, usually less severe than that seen with Dideoxycytidine. The most serious toxicity is acute pancreatitis, which is dose related and uncommonly seen in patients receiving less than 750 mg/day. Alcohol use should be avoided and other drugs associated with pancreatitis used with caution. Diarrhea is a common adverse effect.
Stavudine (d4T), another pyrimidine analog, has in vitro activity similar to azidothymidine’s. It has good oral bioavailability and crosses the blood–brain barrier. In combination with other antiretrovirals, it too has good antiretroviral effect. The most frequent toxicity observed with d4T is peripheral neuropathy. It is a dose-limiting adverse effect, and its occurrence is associated with a prior history of neuropathy or the presence of neurologic symptoms. The symptoms usually resolve when d4t is promptly stopped, and some patients are able to tolerate its reintroduction at a lower dose.
Lamivudine, the (–) enantiomer of 2′-deoxy-3′-thiacytidine (3TC) also has in vitro activity against human immunodeficiency virus-1, including zidovudine-resistant strains. It has high oral bioavailability and is well tolerated. Studies have shown that combination treatment with 3TC and azidothymidine is well tolerated and associated with greater improvement in CD4 counts and viral load than monotherapy. It has been widely studied as part of combination therapy with protease inhibitors.
The investigational drug 1592U89, a potent nucleoside analog in vitro, is currently in clinical trials in antiretroviral naive and experienced patients and available through compassionate access protocols. Abacavir, a potent nucleoside analog in vitro, is the newest NRTI. Hypersensitivity reactions characterized by fever and rash have been treatment-limiting adverse effects.





