Primaquine dose is highly effective to treat malaria as well as Pneumocystis Pneumonia. Read the article to learn more about Primaquine mechanism of action, dosage instructions, side effects, and warnings.

Decisions regarding when to initiate or modify antiretroviral therapy should be guided by monitoring plasma HIV-1 RNA levels (viral load), CD4+ T-cell counts, and the clinical condition of the patient. Although various other surrogate markers and laboratory parameters were used in the past to assess the risk of progression of HIV infection and evaluate efficacy of antiretroviral agents.

Progressive neurodevelopmental deterioration is defined as persistence or progression of deterioration documented on repeated testing as demonstrated by the presence of 2 or more of the following findings: impairment in brain growth, decline in cognitive function documented by psychometric testing, or clinical motor dysfunction. If there is evidence of progressive neurodevelopmental deterioration, the new antiretroviral regimen optimally should include at least one drug that has substantial CNS penetration (e.g., zidovudine or nevirapine).
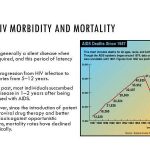
Patient compliance with recommended regimens (even when asymptomatic) is essential to the potential benefits of antiretroviral therapy. Adherence to antiretroviral regimens is an important determinant of both the degree and duration of virologic suppression. Excellent adherence has been shown to increase the likelihood of sustained virologic control, which is important for reducing HIV-associated morbidity and mortality. Poor adherence has been shown to increase the likelihood of virologic failure and can lead to the development of resistance and limit the effectiveness of antiretroviral therapy.

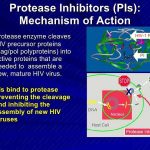
While further study is needed, data are accumulating regarding pharmacokinetic interactions among the various antiretroviral agents, especially those involving the HIV protease inhibitors and NNRTIs, and the need for dosage adjustments as a result of these interactions. While some pharmacokinetic interactions between antiretroviral agents can be used for therapeutic advantage (e.g., use of low-dose ritonavir to boost plasma concentrations of some other HIV protease inhibitors), other interactions can result in suboptimal drug concentrations and reduced therapeutic effects and should be avoided. The pharmacokinetic interaction between ritonavir and other HIV protease inhibitors is now used for therapeutic advantage in various antiretroviral regimens.
The fact that hyperglycemia, new-onset diabetes mellitus, exacerbation of preexisting diabetes mellitus, and diabetic ketoacidosis have occurred in HIV-infected individuals receiving HIV protease inhibitors should be considered when these drugs are used during pregnancy. Because pregnancy is itself a risk factor for hyperglycemia and it is not known whether use of an HIV protease inhibitor exacerbates this risk, glucose concentrations should be monitored closely in pregnant women receiving these drugs and these women should be advised about the warning signs of hyperglycemia and diabetes (e.g., increased thirst and hunger, unexplained weight loss, increased urination, fatigue, dry or itchy skin).