Essentials of Diagnosis
- Most common infections include pneumonia, meningitis, sinusitis, and otitis media.
- Predisposing factors include extremes of age (ie, infants and elderly) and underlying host defects in antibody, complement, and splenic function.
- Transmission is human to human both in the community and nosocomially.
- Pneumonia: productive cough, fever, chills, sweats, and dyspnea; lobar or segmental consolidation on chest x-ray; lancet-shaped gram-positive diplococci on Gram stain of sputum or growth of S pneumoniae from sputum, blood, or pleural fluid.
- Meningitis: headache, stiff neck, fever, chill, and photophobia; nuchal rigidity, Kernig’s or Brudzinski’s sign; lancet-shaped gram-positive diplococci on Gram stain of cerebrospinal fluid (CSF), growth of S pneumoniae from CSF or blood, or positive counter immunoelectrophoresis or latex agglutination of CSF.
General Considerations
Epidemiology
S pneumoniae may exist in humans as either a nasopharyngeal colonist or as a pathogen in one of many clinical syndromes (Box 1). Although S pneumoniae has been found in other mammals, humans are thought to be the principal reservoir. As a colonist, S pneumoniae is found in up to 40% of children and 10% of adults. Infants typically acquire their first colonizing isolate at 6 months. Intermittent colonization then occurs throughout childhood and adult years. There is a seasonal pattern to colonization, with the highest prevalence occurring in winter. Pneumococci are transmitted from human to human by extensive direct contact with secretions harboring the organism. Persons residing in environments that promote such extensive close contact, such as day care centers, prisons, homeless shelters, and military barracks, have historically been at risk for pneumococcal infection.
In the United States, S pneumoniae is estimated to account for 500,000 cases of pneumonia, 50,000 cases of bacteremia, 3000 cases of meningitis, and 7 million cases of acute otitis media. Of these syndromes, bacteremia (with or without a site of primary infection such as pneumonia or meningitis) has the most clearly elucidated epidemiology because of its precise blood culture-based definition. The incidence of bacteremia is strongly age related. A surveillance study in South Carolina estimated the incidence of bacteremia among infants, young adults, and elderly (age = 70) to be 160, 5, and 70 cases per 100,000 persons, respectively. African-Americans, American Indians, and Alaskan Natives are at highest risk for bacteremia. As is true for S pneumoniae colonization, there is a distinct seasonality to bacteremia with peaks coming at midwinter. Overall, pneumococcal disease accounts for ~ 40,000 annual deaths in the United States. The case fatality rate for bacteremia is 15-20%.
In addition to the numerical incidence of pneumococcal disease, it is instructive to look at the prominent role S pneumoniae plays in several common syndromes. In a variety of studies that prospectively identify causes of community-acquired pneumonia, S pneumoniae is routinely the most common detectable agent. For example, in patients who require hospitalization for pneumonia, S pneumoniae accounts for up to one-third of cases. Aside from areas of the world experiencing meningococcal epidemics, S pneumoniae is the most common organism of meningitis in adults and now, with the advent of Haemophilus influenzae type b immunization, is also most common in children. One-third to one-half of all cases of acute otitis media with an identifiable etiology is caused by S pneumoniae.
Any of the syndromes caused by S pneumoniae can occur in normal hosts, but frequently one or more predisposing conditions exist. The best characterized of these are underlying host immunologic defects in antibody, complement, and splenic function. Because of the importance of antibodies in the opsonization of the encapsulated pneumococcus, individuals with defective antibody function (eg, congenital agammaglobulinemia, common variable hypogammaglobulinemia, selective immunoglobulin-G subclass deficiency, multiple myeloma, chronic lymphocytic leukemia, or lymphoma) are at significant risk for invasive infections. Various early complement deficiencies as well as either congenital or acquired asplenia (eg, iatrogenic or sickle cell disease) also convey substantial risk. Asplenic patients deserve special note because of the rapidity at which they can clinically deteriorate from unchecked bacteremia.
In contemporary urban settings, HIV infection is responsible for a large burden of pneumococcal disease. HIV disease, through a variety of host defense defects, places infected persons at ~ 200-fold-greater risk for invasive pneumococcal infection. HIV-infected patients who are African-American, who have a CD4 lymphocyte count < 200 × 106/liter, or who have a history of pneumonia have particularly elevated risk. The presence of pneumococcal bacteremia in patients with no clinically apparent host immunodeficiency should prompt the clinician to obtain patient consent for HIV testing.
Various other chronic illnesses, such as alcoholism, cirrhosis, renal insufficiency, nephrotic syndrome, chronic pulmonary disease, congestive heart failure, and diabetes, place patients at significant risk for pneumococcal disease. Likewise, patients who have received organ or bone marrow transplants or who are being treated with alkylating agents, antimetabolites, or glucocorticoids are also at risk. Finally, a particular anatomic defect, basilar skull fracture, carries risk for pneumococcal meningitis.
Microbiology
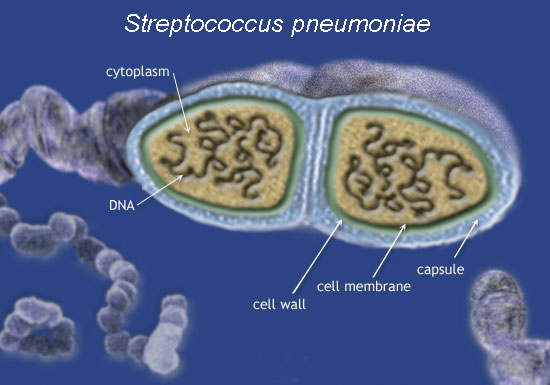
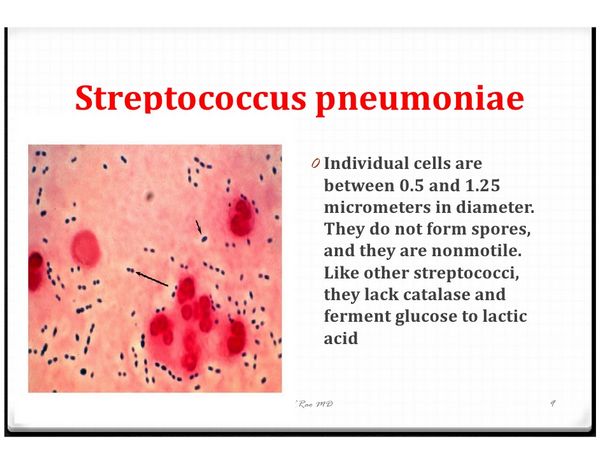
S pneumoniae is a gram-positive coccus, but typically the organism grows as pairs in what is termed a lancet-shaped diplococcus. In truth, this shape represents two lancets joined on end, pointing away from each other. In liquid medium, pneumococci may also be found growing in chains. On blood agar plates, pneumococcal colonies are often umbilicated (ie, have central dimpling) and are sometimes grossly mucoid. They are surrounded by a greenish zone, known as a-hemolysis, which is the result of hemoglobin breakdown by pneumococcus-produced a-hemolysin. In the clinical microbiology laboratory, a-hemolysis is the first clue in distinguishing S pneumoniae from a variety of other species in a mixed flora sample. In addition to a-hemolysis, the microbiology laboratory uses catalase negativity, solubility in bile salts, and susceptibility to optochin to identify S pneumoniae (Table 1).
S pneumoniae is easy to identify by the above criteria when it grows in normally sterile body sites (eg, blood or pleural fluid). Detection in a mixed-flora environment such as sputum, however, requires a skilled and persevering clinical microbiologist. Given the abundance of other a-hemolytic oral species, it is incumbent on the microbiologist to look carefully for the characteristic umbilicated or mucoid appearance of S pneumoniae and to subculture a number of a-hemolytic colonies. The clinician can be motivating in this endeavor by communicating the patient’s clinical presentation to the microbiologist and by carefully reviewing the Gram stain of the original specimen for the presence of lancet-shaped diplococci.
Once called Diplococcus pneumoniae, S pneumoniae is now known to belong to the Streptococcus genus. Like most other members of this genus, S pneumoniae is non-spore forming and nonmotile. Unlike many streptococcal species, S pneumoniae does not have a Lancefield serogroup. Yet, within the S pneumoniae species, there are 90 identified serotypes. Two numbering schemes for serotyping exist. In the American system, serotypes are numbered starting from 1 to 90 in the order in which they were first described. In the more widely used Danish system, serotypes are grouped based on antigenic similarities and given both a number and a letter (eg, 19A, 19B, and 19C). The basis of the serotype is antigenic differences in the polysaccharides that constitute the pneumococcal external capsule. Almost all clinical strains, with the uncommon exception of some isolates from conjunctivitis, have a capsule. In the microbiology laboratory, the interaction of serotype-specific antibody with capsule results in the enhancement of the microscopic appearance of the capsule, which is known as the quellung reaction. Although serotyping is no longer an important clinical tool, it remains useful for epidemiologic studies; surveys of the most prevalent serotypes found in invasive infections have informed the composition of the current polyvalent vaccine.
Whereas the external capsule is a distinguishing feature of S pneumoniae, its cell wall is similar to that of other streptococci. Multilayered peptidoglycan, a heteropolymer of repeating N-acetylglucosamine and N-acetyl muramic acid, is the principal component. Peptides, attached to N-acetyl muramic acid, are cross-linked by trans- and carboxypeptidases to provide structure to the cell wall. These peptidases are also known as penicillin-binding proteins because they are the binding sites for a variety of ß-lactam antibiotics. The covalent binding of ß-lactams inactivates these enzymes resulting in the eventual demise of the organism. Teichoic acid (ribotol- or glycerol-phosphate polymers) is the other principal component of the cell wall and is often covalently bound to peptidoglycan. Lipoteichoic acid exists as a component of both the cell membrane and cell wall. There are other minor components of the cell wall, some of which are common to all streptococci, and some of which, like C-substance (a polysaccharide), are unique to S pneumoniae. Various soluble products are produced by S pneumoniae and are discussed in the next section.
Pathogenesis
Diseases caused by S pneumoniae are principally the result of an exuberant inflammatory response that the host mounts to the organism. The critical factor in the success of the pneumococcus is its ability to evoke this response but simultaneously, at least until specific anticapsular antibody is produced, to evade nonspecific attempts at phagocytosis. Highly destructive toxins, analogous to those produced by Streptococcus pyogenes or gram-negative species, are not present although some toxins are produced and play a secondary role. Although details vary slightly depending on the organ system involved, the pathogenic progression consists of nasopharyngeal adherence and colonization, spread to adjacent organs with local replication, evasion of nonspecific clearance mechanisms, and finally activation of the host inflammatory reaction.
The initiating event in all cases is nasopharyngeal colonization, in which bacterial surface adhesins join to epithelial cell receptors that contain the disaccharide GlcNAcß1-4Gal. After pneumococci are established in the nasopharynx, they may then gain entry into and replicate in contiguous structures such as the sinuses, eustachian tubes, or bronchi. They may also penetrate the nasopharyngeal mucosa, even without a clinically demonstrable focus of infection, and achieve access to the systemic circulation via the cervical lymphatics. Entry into the lungs is limited in normal hosts by a functioning glottis and larynx, but entry is enhanced when these mechanisms are disturbed, as seen with alcohol intoxication, convulsions, anesthesia, or stroke.
Even after pneumococci gain entry to the above structures, infection rarely occurs because they are typically cleared in the normal host by nonspecific mechanisms (eg, mucociliary motion or cough reflex). Replication can proceed, however, when normal clearance is impaired, as seen, for example, when eustachian tubes or sinus orifices are congested (eg, from viral infection or allergy) or bronchial clearance is altered (eg, from chronic effects of smoking or acute effects of viral infection). In the lung, various mechanisms are now being described that may enhance pneumococcal adherence and replication. These include the finding that pneumococci bind to immobilized fibronectin that is exposed during tissue injury and that cytokines may induce pneumocytes to express the receptor for platelet-activating factor—a receptor that also binds pneumococcal C-substance.
Once established in its target organs, S pneumoniae is able to replicate unimpeded because of its ability to evade nonspecific phagocytosis by polymorphonuclear cells. The external capsule provides this crucial protection. As such, the capsule is an essential factor in the virulence of the organism. The exact mechanism of this protective effect is not known; it is likely a combination of the lack of capsular receptors on phagocytes, repellent electrochemical forces on the capsule, and the ability of the capsule to mask opsonizers such as antibody and complement. Whatever the exact mechanism, S pneumoniae organisms that are genetically engineered to lack capsules are avirulent.
Once it begins replicating, S pneumoniae causes disease by evoking an intense inflammatory reaction. Both the peptidoglycan and teichoic acid components of the cell wall and capsular polysaccharide can activate the alternative pathway of complement. Nonspecific antibodies to cell wall polysaccharides (formed to ubiquitous streptococcal species) are able to activate complement by the classical pathway. Together, these means of activating complement result in the attraction of numerous leukocytes and abundance of exudative fluid. In the lungs, this accumulation of pneumococci and inflammatory material leads to consolidation of alveoli, radiolucency, and impaired gas exchange—the defining characteristics of pneumonia. Although abscess formation is rare, the expanding volume of infected material can spread to uninvolved areas via Kohn’s pores. Extensive direct spread can result in empyema or pericardial infection. If the infection is not contained, pneumococci can spread via lymphatics to hilar lymph nodes, the thoracic duct, and finally into the systemic circulation where metastatic infection may occur in the meninges, peritoneum, joints, or endocardium.
As mentioned, S pneumoniae may gain access to the subarachnoid space via hematogenous spread and choroid seeding from either a nasopharyngeal or pulmonary focus. Less commonly, meninges may become infected via direct extension from an infected sinus or middle ear. As in the lungs, an intense inflammatory response is evoked by the presence of pneumococci in the subarachnoid space. Interleukin-1 and tumor necrosis factor, released by macrophages responding to the insult, play a prominent role and contribute to increased blood-brain barrier permeability. The end result is mounting intracranial pressure and ultimately diminished blood flow to the brain.
Before the advent of antibiotics, this pathogenic process could be arrested by the development of specific anticapsular antibody. This typically occurs at 5-8 days after the onset of infection. Hence, outcome depended on whether antibody production occurred before the patient succumbed to the severe manifestations of infection. In the contemporary era, it is now appreciated that the pathogenic process can also be arrested by the administration of antibiotics. However, the persistently high case fatality rate associated with pneumococcal disease (especially bacteremia) despite antibiotic administration bespeaks the continued contributory role of host defenses in successfully responding to infection.
S pneumoniae produces several toxins that are currently thought to play a secondary role in pathogenesis. Pneumolysin is a toxin with two functions. It can insert into cell membranes of polymorphonuclear cells and ciliated epithelium, thereby inhibiting their function, and it can activate the classical complement pathway. Other products such as pneumococcal surface protein, hemolysin, and autolysin are also contributory because mutants lacking these typically are less virulent. Autolysin in particular is important because autolysis of organisms may enhance the release and expression of a variety of proinflammatory factors (eg, cell wall components) that were discussed above.
CLINICAL SYNDROMES
Table 1. Clues to diagnosis of Streptococcus pneumoniae infection
- Gram-positive coccus; grows as lancet-shaped diplococci or in chains
- Grows readily in conventional blood culture or solid media, but fastidious nature necessitates rapid transport of clinical material to microbiology laboratory for processing
- a-Hemolytic (greenish) on solid media; colonies are often umbilicated or mucoid
- Catalase-negative
- Bile salt soluble
- Susceptible to optochin
- Counter immunoelectrophoresis or latex agglutination may be useful in evaluation of cerebrospinal fluid that was obtained after antibiotics were administered
BOX 1. Syndromes Caused by Streptococcus pneumoniae
More Common
- Pneumonia (with or without bacteremia)
- Acute sinusitis
- Otitis media
- Meningitis
- Primary bacteremia
Less Common
- Empyema
- Septic arthritis
- Peritonitis
- Endocarditis
- Pericarditis
- Cellulitis
- Endometritis
- Osteomyelitis
- Brain abscess
More common in children than adults.
BOX 2. Clinical Findings in Pneumococcal Pneumonia
Symptoms
Signs
Laboratory Tests
More Common
- Fever
- Chills
- Sweats
- Cough
- Rusty sputum production
- Dyspnea
- Fever, tachycardia, tachypnea
- Consolidative findings on chest exam
- Hypoxemia
- Elevated leukocyte count
Less Common
- Pleuritic chest pain
- Nausea, vomiting, diarrhea
- Confusion
- Hypothermia
- Hypotension
- Cyanosis
- Oral herpes lesions
- Pleural rub
- Murmur
- Altered mental status
- Low serum bicarbonate, anion gap
- Elevated total bilirubin and lactate dehydrogenase
- Nuchal rigidity
- Abdominal distension and absent bowel sounds
- Radiographic and microbiologic findings are discussed in the text.
- Many of these findings represent the presence of concomitant pneumococcal disease in other organ systems.
BOX 3. Definitive Treatment of Streptococcus pneumoniae Pneumonia Based on Penicillin Susceptibility
Susceptible (MIC <0.1 µg/mL)
Intermediate Resistance
(MIC 0.1-1.0 µg/mL)
Highly Resistant (MIC = 2.0 µg/mL)
Children
Adults
Children
Adults
Children
Adults
First Choice
Penicillin V (7 mg/kg orally every 6 h — maximum 500 mg/dose)
OR Penicillin G (16,500 U/kg IV every 4 h — maximum 1 million U/dose)
Penicillin V (500 mg orally four times daily)
OR Penicillin G (1 million U IV million every 4 h)
Amoxicillin (13.3 mg/kg orally three times daily — maximum 500 mg/dose)
OR Penicillin G (33,000 U/kg IV every 4 h — maximum 2 million U/dose)
Amoxicillin (500 mg orally three times daily)
OR Penicillin G (2 million U IV every 4 h)
Cefpodoxime2 (5 mg/kg orally every 12 h — maximum 200 mg/dose)
OR Ceftriaxone2 (25 mg/kg IV every 12 h — maximum 1 g/dose)
Cefpodoxime2 (200 mg orally twice daily) OR Ceftriaxone2 (1 g IV every 12 h)
Second Choice
Amoxicillin (13.3 mg/kg orally three times daily — maximum 500 mg/dose)
OR Cefazolin (20 mg/kg IV every 8 h — maximum 1 g/dose)
Amoxicillin (500 mg orally three times daily)
OR Cefazolin (1 g IV every 8 h)
Cefuroxime axetil (15 mg/kg orally twice daily — maximum 500 mg/dose)
OR Cefazolin (20 mg/kg IV every 8 h — maximum 1 g/dose)
Cefuroxime axetil (500 mg orally twice daily)
OR Cefazolin (1 g IV every 8 h)
Cefotaxime2 (25 mg/kg IV every 6 h — maximum 1 g/dose)
Cefotaxime2 (1 g IV every 6 h)
Penicillin Allergic
Erythromycin (7.5 mg/kg orally every 6 h or 5 mg/kg IV every 6 h — maximum 500 mg/dose)
OR Clindamycin 4 mg/kg orally four times daily — maximum 300 mg/dose or 8.3 mg/kg IV every 8 h — maximum 300 mg/dose)
Erythromycin (500 mg orally or IV every 6 h)
OR Clindamycin (300 mg orally four times daily or 600 mg IV every 8 h)
Clindamycin (4 mg/kg orally four times daily — maximum 300 mg/ dose or 8.3 mg/kg IV every 8 h — maximum 600 mg/dose)
Clindamycin (300 mg orally four times daily or 600 mg IV every 8 h)
Vancomycin (10 mg/kg IV every 6 h)
Vancomycin (1 g IV every 12 h)
- Choices for oral and parenteral therapy are given. The use of oral vs parenteral therapy depends on the patient’s clinical course and gastrointestinal function. All dosages assume normal renal function.
- Assuming in vitro susceptibility. If not susceptible to cephalosporins, a suitable fluoroquinolone (such as sparfloxacin or levofloxacin) or vancomycin must be used.
BOX 4. Clinical Findings in Pneumococcal Meningitis
Symptoms
Signs
Laboratory Tests1
More Common
- Headache
- Stiff neck
- Fever
- Chills
- Sweats
- Photophobia
- Fever, tachycardia
- Nuchal rigidity
- Kernig’s and Brudzinski’s signs
- Elevated leukocyte count
- Increased intracranial pressure (>180 mm H2O)
- CSF pleocytosis (500 to 10,000 cells/mm3, with polymorphonuclear cell predominance)
- Elevated CSF protein (100-500 mg/dL)
- Depressed CSF glucose (<40 mg/dL)
Less Common
- Cough
- Earache or facial pressure/postnasal discharge
- Confusion
- Hypotension
- Papilledema
- Focal neurologic deficits
- Hyperesthesia
- Altered mental status
- Consolidative findings on chest exam
- Purulent discharge from nose or ear; bulging tympanic membrane
- Hypoxemia
- Radiographic and microbiologic findings are discussed in the text.
- Many of these findings represent the presence of concomitant pneumococcal disease in other organ systems.
BOX 5. Definitive Treatment of Streptococcus pneumoniae Meningitis Based on Penicillin Susceptibility
Susceptible (MIC < 0.1 µg/mL)
Intermediate or Highly Resistant
(MIC = 0.1 µg/mL)
Children
Adults
Children
Adults
First Choice
Penicillin G (50,000 U/kg IV every 4 h — maximum 4 million U/dose)
Penicillin G (4 million U IV every 4 h)
Ceftriaxone2 (50 mg/kg IV every 12 h — maximum 2 g/dose)
Ceftriaxone2 (2 g IV every 12 h)
Second Choice
Ceftriaxone (50 mg/kg IV every 12 h — maximum 2 g/dose) OR Cefotaxime (50 mg/kg IV every 6 h; maximum 2 g/dose)
Ceftriaxone (2 g IV every 12 hours OR Cefotaxime (2 g IV every 6 h)
Cefotaxime2 (50 mg/kg IV every 6 h — maximum 2 g/dose)
Cefotaxime2 (2 g IV every 6 h)
Penicillin Allergic
Vancomycin3 (15 mg/kg IV every 6 h)
Vancomycin3 (1 g IV every 12 h)
Vancomycin (15 mg/kg IV every 6 h)
Vancomycin (1 g IV every 12 h)
- All dosages assume normal renal function. Adjunctive dexamethasone should be given to children at least 15 min before the first dose of antibiotics. See text for discussion of steroid use in adults.
- Assuming in vitro susceptibility. If not susceptible to ceftriaxone or cefotaxime, vancomycin must be used. Even if isolates are resistant to ceftriaxone and ceftotaxime, one of these agents should be considered for use in addition to vancomycin.
- Either cefotaxime or ceftriaxone may be considered if the penicillin allergy is known not to be anaphylactic immediate type. The penetration of these agents is more reliable than vancomycin.
BOX 6. Control of Invasive Streptococcus pneumoniae Infections
Prophylactic Measures
23-valent polysaccharide vaccine is recommended for the following persons1:
- Aged = 65 y
- Aged 2-64 y with chronic illnesses such as chronic cardiovascular disease (coronary artery disease, congestive heart failure, and cardiomyopathies), chronic pulmonary disease (chronic obstructive pulmonary disease but not isolated asthma), diabetes mellitus, alcoholism, chronic liver disease (including cirrhosis), cerebrospinal fluid leaks, and functional or anatomic asplenia (including sickle cell disease)
- Aged 2-64 y living in special environment or social settings, such as Alaskan natives and certain American Indian populations as well as residents of nursing homes and other long-term-care facilities
- Aged 2-64 y who are immunocompromised, including those with HIV infection, leukemia, lymphoma, Hodgkin’s disease, multiple myeloma, generalized malignancy, chronic renal failure, or nephrotic syndrome; those receiving immunosuppressive chemotherapy (including corticosteroids); and those who have received an organ or bone marrow transplant
Heptavalent conjugate vaccine is recommended for the following persons:
- Aged <2 y and high-risk children between 2 and 5 years of age
Daily oral penicillin V is recommended for children with functional (eg, sickle cell disease) or anatomic asplenia
Isolation Precautions
None required
Other Syndromes
Primary pneumococcal bacteremia (ie, no identifiable anatomic focus) is common in children but infrequent in adults. S pneumoniae is the major pathogen of otitis media in children, being responsible for between 33% and 50% of all cases in which an etiologic agent can be identified. It is also one of the two most important (along with H influenzae) agents implicated in acute sinusitis. Empyema, typically a complication of pneumonia but rarely a primary diagnosis, has been discussed above. Less common syndromes include endocarditis and pericarditis, septic arthritis and osteomyelitis, peritonitis, endometritis, brain abscess, and cellulitis. Most of these can occur as either a primary infection or as a complication of an initiating clinical focus (eg, pneumonia). (Refer to other chapters in this text for a complete description of these syndromes, including a discussion of empiric and definitive treatments.)
The principles discussed above regarding the various levels of certainty in the attribution of a syndrome to S pneumoniae, knowledge of local prevalence of drug-resistant isolates, and the importance of the antimicrobial susceptibility pattern of the isolate (if obtained) are similarly pertinent when determining empiric and definitive therapy for other syndromes.
Prevention & Control
A polyvalent vaccine against S pneumoniae, manufactured by both Merck (Pneumovax 23) and Lederle (Pnu-Immune 23), contains 25 ug of capsular polysaccaride from each of the 23 most common serotypes responsible for invasive pneumococcal infections in the United States. Vaccine serotypes represent = 85-90% of the isolates responsible for invasive infections. It is important to note that the six serotypes that are the most frequent causes of penicillin-resistant infections are included. The vaccine has been shown to be effective for the prevention of invasive pneumococcal disease (eg, bacteremia with or without pneumonia) in immunocompetent adults aged = 65 years and in persons aged = 2 years with chronic illnesses such as diabetes mellitus, alcoholism, cirrhosis, chronic pulmonary disease, coronary artery disease, and congestive heart failure (Box 6). Although convincing data on vaccine effectiveness in other immunocompromised populations (eg, HIV infection in persons from developed countries [data from sub-Saharan Africa show no vaccine efficacy], leukemia, Hodgkin’s disease, multiple myeloma, and chronic renal failure) have not been presented, methodologic constraints or frank absence of dedicated studies has precluded excluding a protective role. Therefore, vaccination is generally recommended for these populations.
For infants < 2 years old, for whom the 23-valent vaccine is ineffective, a heptavalent vaccine (Prevnar, Wyeth-Lederle) containing the capsular polysaccharides of serotypes 4, 6B, 9V, 14, 18C, 19F, and 23F conjugated to a nontoxic diphtheria protein has been found effective in the prevention of invasive pneumococcal disease (Box 6). A series of immunizations with this conjugate vaccine in infants 2, 4, and 6 months old followed by a fourth dose when these infants are 12-15 months old is now recommended for all newborns. The vaccine is also recommended for high-risk children (eg, those with sickle cell disease or asplenia) between 2 and 5 years of age.
The Advisory Committee on Immunization Practices of the US Public Health Service has recently published updated recommendations on revaccination. This committee does not recommend routine revaccination but suggests that individuals = 2 years old who are at highest risk for serious pneumococcal infection and for whom rapid declines in antibody titers are known to occur should receive a second vaccination 5 years after the initial vaccine. These individuals include patients with HIV disease, functional or anatomic asplenia, leukemia, lymphoma, Hodgkin’s disease, multiple myeloma, generalized malignancy, chronic renal failure, nephrotic syndrome, or transplants and those receiving immunosuppressive therapy. It is also recommended that individuals = 65 years old receive a second vaccination 5 years after the initial vaccine, provided that the first vaccine was administered when they were < 65 years old.
In children with either functional (eg, sickle cell disease) or anatomic asplenia, chemoprophylaxis with daily oral penicillin V provides another means of prevention and is also recommended.
Although human-to-human transmission of S pneumoniae is common, illness among contacts is very infrequent in nonepidemic settings. Accordingly, isolation of hospitalized patients or immunization of contacts is not recommended.



