Great overlap occurs among the clinical manifestations of the pathogens associated with acute community-acquired pneumonia. However, constellations of symptoms, signs, and laboratory findings serve to narrow the possibilities. By developing an ability to focus on a few pathogens or to identify a specific pathogen, clinicians can better predict the clinical course of pneumonia and can narrow antibiotic coverage.
Streptococcus pneumoniae
Pathogenesis
Pathogenic strains of S. pneumoniae have a thick capsule that prevents PMN binding and that blocks phagocytosis. Certain capsular types (1, 3, 4, 7, 8, and 12 in adults, and 3, 6, 14, 18, 19, and 23 in children) account for most pneumonia cases. Type 3 has the thickest polysaccharide capsule, and it is the most virulent strain, being associated with the worst prognosis. Immunoglobulins that specifically recognize the capsule are able to link the bacterium to the PMN surface through Fc receptors, enabling polymorphonuclear leukocytes and macrophages (classified as phagocytes) to efficiently ingest and kill the pneumococci. The complement product C3b enhances phagocytosis of the bacteria by the same mechanism. Immunoglobulins and C3b are called “opsonins,” which are products that enhance foreign particle ingestion by phagocytes.
In addition to its polysaccharide capsule, S. pneumoniae possesses a number of other virulence factors that enhance adherence to epithelial cells, resist phagocytosis, and activate complement. S. pneumoniae does not produce significant quantities of proteases, and disease manifestations are primarily the consequence of the host’s inflammatory response. As a result, permanent tissue damage is rare, and spread of the disease across anatomic boundaries, such as lung fissures, is uncommon.
About Treatment and Outcome of Pneumonia
- Treatment must be instituted within 4 hours of diagnosis.
- Delays are associated with increased mortality.
- Appropriate triage should be guided by the CURB-65 classification.
- Empiric therapy depends on the patient and disease characteristics:
- Outpatient with no comorbidity and no previous antibiotics. Use a macrolide (azithromycin or clarithromycin). If previous antibiotics or elderly nursing home patient, add a p-lactam antibiotic, or use a respiratory fluoroquinolone.
- Hospitalized patient. Use a 3rd-generation cephalosporin (ceftriaxone or cefotaxime) combined with a macrolide (azithromycin or clarithromycin). If Pseudomonas is a concern, use piperacillin-tazobactam, imipenem, or meropenem.
- Aspiration outpatient. Use penicillin or clindamycin.
- Aspiration inpatient. Use a 3rd-generation cephalosporin or a respiratory fluoroquinolone plus metronidazole; or use ticarcillin-clavulanate or piperacillin-tazobactam.
- Using chest radiographs to monitor improvement is not recommended. (They can take several weeks to clear.) They are useful for documenting worsening of disease or development of complications.
- Mortality ranges from 2% to 30%. Mortality higher with age more than 65 years, neoplas-tic disease, liver disease, congestive heart failure, cerebrovascular accident, and renal disease.
Prevalence and Predisposing Factors
S. pneumoniae remains the most common cause of acute community-acquired pneumonia; it represents two thirds of the cases in which a specific pathogen is identified. Because opsonins are required for efficient phagocytosis of the encapsulated organism, patients with hypogammaglobulinemia and multiple myeloma are at increased risk for developing this infection, as are patients with deficiencies in complement (Cl, C2, C3, C4). Patients with HIV infection also have defects in antibody production, and they have a higher incidence of pneumococcal infection.
About the Pathogenesis of Streptococcus pneumoniae
- The thick outer capsule blocks phagocytosis. Type 3 has the thickest capsule.
- Immunoglobulins and complement are important opsonins that allow phagocytes to ingest invading pneumococci.
- Strep, pneumoniae does not produce protease and seldom destroys lung parenchyma.
- It does not cross anatomic barriers such as lung fissures.
- Disease manifestations are caused primarily by the host’s inflammatory response to the organism.
About Streptococcus pneumoniae Prevalence and Predisposing Factors
- S. pneumoniae is the most common form of community-acquired bacterial pneumonia.
- The risk is higher in patients with deficiencies in opsonin production:
- Hypogammaglobulinemia
- Complement deficiency
- HIV infection
- Splenic dysfunction increases the risk of fatal pneumococcal bacteremia.
- Risk is increased in patients with chronic diseases:
- Cirrhosis
- Alcoholism
- Nephrotic syndrome
- Congestive heart failure
- Chronic obstructive pulmonary disease
About Clinical Manifestations and Diagnosis of Pneumococcal Pneumonia
- Three classic features may be found:
- Abrupt onset accompanied by a single rigor
- Rusty-colored sputum
- Pleuritic chest pain
- Sputum Gram stain is often helpful: more than 10 gram-positive lancet-shaped diplococci per high-power field indicate pneumococcal pneumonia.
- Sputum culture is insensitive; specimens (alpha hemolytic, optochin sensitive) should be plated quickly.
- Blood samples for culture should always be drawn; up to 25% may be positive.
- A urine pneumococcal antigen test may prove helpful, but may be positive in patients who are simply colonized with Streptococcus pneumoniae.
- A chest radiograph shows a classical lobar pattern; small pleural effusions are common, true empyema rare. Abnormalities persist for 4 to б weeks after cure.
Patients with splenic dysfunction have a higher risk of overwhelming S. pneumoniae sepsis because the spleen plays a vital role in clearing this bacteria from the bloodstream, particularly in the absence of specific anti-pneumococcal capsule antibody. Other chronic diseases, including cirrhosis, nephrotic syndrome, congestive heart failure, chronic obstructive pulmonary disease, and alcoholism, are also associated with greater risk of pneumococcal infection.
Unique Clinical Characteristics
Classically pneumococcal pneumonia has a very abrupt onset that begins with a single severe rigor. Because S. pneumoniae invasion of the lung leads to capillary leakage of blood into the alveolar space, sputum can become rusty in color. Furthermore, pneumococcal infection frequently infects the peripheral lung and spreads quickly to the pleura. As a result, pleuritic chest pain is a common complaint.
Diagnosis
Sputum Gram Stain — A careful analysis of the sputum is best performed by a knowledgeable physician. Areas with significant numbers of polymorphonuclear leukocytes per high-power field and a predominance of gram-positive lancet-shaped diplococci suggest the diagnosis. A finding of pneumococci within the cytoplasm of a PMN strongly supports invasive infection.
Sputum Culture — S. pneumoniae is catalase negative, bile soluble, and, like S. viridans, demonstrates alpha (green) hemolysis on blood agar plates. The propensity of normal mouth flora, in particular S. viridans, to overgrow frequently interferes with the identification of S. pneumoniae. The optochin disk inhibits growth ofS. pneumoniae, but not of S. viridans, and this test is used to differentiate the two organisms. Another problem with sputum culture arises from the fact that S. pneumoniae can be present as normal mouth flora in up to 60% of healthy people. A positive sputum culture in the absence of a positive Gram stain or a positive blood culture may therefore simply represent contamination of the sputum with saliva.
Blood Cultures — Some reports have claimed that 25% of patients with pneumococcal pneumonia develop positive blood cultures; however, the denominator required to calculate this percentage is uncertain. Even in the absence of a positive sputum Gram stain, a positive blood culture in combination with the appropriate symptoms and Chest X-Ray findings is interpreted as true infection. A urine test for pneumococcal polysac-charide antigen is available and is positive in 80% of adults with bacteremia.
The Chest X-Ray usually reveals a single area of infiltration involving one or more segments of a single lobe. Involvement of the entire lobe is less common. This organism respects the confining fissures of the lung and rarely extends beyond those boundaries, which explains the classical lobar radiologic pattern.
Air bronchograms are found in a few cases. This radiologic finding is the consequence of the alveoli filling with inflammatory fluid and outlining the air-containing bronchi. When found, bronchograms are associated with a higher incidence of bacteremia.
Pleural fluid may be detected in up to 40% of cases. In most instances, the volume of fluid is too small to sample by thoracentesis, and if antibiotic treatment is prompt, only a small percentage go on to develop true empyema.
The radiologic improvement of pneumococcal pneumonia is slow. Despite rapid defervescence and resolution of all symptoms, radiologic changes often persist for 4 to 6 weeks. If the patient is improving clinically, follow-up CXRs are therefore not recommended during this period.
Treatment and Outcome
In the early antibiotic era, S. pneumoniae‘was highly sensitive to penicillin [minimum inhibitory concentration (minimum inhibitory concentration)< 0.06 g/mL]. However, since the late 1990s,
isolates in the United States have become increasingly resistant, with 40% demonstrating intermediate resistance (minimum inhibitory concentration = 0.1-1 µg/mL), and a small percentage demonstrating high-level resistance (minimum inhibitory concentration > 2 µg/mL). In some areas of Europe and South Africa, higher percentages of resistant strains have been observed. In the Netherlands and Germany, where strictly limited antibiotic use is the standard of care, the prevalence of resistant strains is lower.
Currently, many intermediate strains remain sensitive to the 3rd-generation cephalosporins ceftriaxone and cefotaxime (minimum inhibitory concentration < 1 µg/mL); however, resistance to these antibiotics is increasing. For intermediately resistant strains, amoxicillin is more active than is penicillin VK, and amoxicillin is therefore the preferred oral antibiotic. Because penicillin resistance results from a decrease in the affinity of penicillin-binding proteins, intermediate (but not high-level) resistance can be overcome by raising the concentration of penicillin.
With the exception of the Central nervous system, where the blood-brain barrier limits antibiotic penetration, standard doses of penicillin are effective in curing infections attributable to intermediately resistant pneumococci. Penicillin resistance is usually associated with resistance to many other classes of antibiotics, including the tetracyclines, macrolides, and clindamycin. Imipenem is also inactive against highly resistant strains. The respiratory fiuoro-quinolones that possess good gram-positive activity (lev-ofloxacin, gatifloxacin, moxifloxacin) and vancomycin usually retain excellent activity against all resistant strains. Several cases of pneumonia attributable to levofloxacin-resistant S. pneumoniae have recendy been reported; however, the overall percentage of pneumococcal strains that are resistant to fluoroquinolones remains low.
Treatment Recommendations
For penicillin-sensitive strains, penicillin G or amoxicillin remain the preferred treatment. Ceftriaxone is also effective. If the patient fails to improve within 48 hours, the possibility of a resistant strain must be considered, and coverage with a respiratory fluoroquinolone is recommended. For cases in which meningitis is suspected, a fluoroquinolone should not be used because of poor penetration of the cerebrospinal fluid, and the patient should be covered with vancomycin. In the penicillin-allergic patient, a respiratory fluoroquinolone can be used.
In the pre-antibiotic era, the mortality rate for pneumococcal pneumonia was 20% to 40%. In the antibiotic era, the mortality rate was reduced to approximately 5%. Prognosis is adversely influenced by
- Age (patients above 65 years of age and infants have worse outcomes)
- Delayed treatment
- Infection with capsular type 2 or 3
- Involvement of more than one lobe of the lung
- white blood cell count less than 6000/mm3
- Bacteremia, shock, or the development of meningitis Jaundice
- Pregnancy
- Presence of other underlying diseases (heart disease, cirrhosis, diabetes)
- Alcohol intoxication
About the Treatment, Outcome, and Prevention of Pneumococcal Pneumonia
- A significant percentage of Streptococcus pneumoniae are resistant to penicillin:
- 25% to 35% are intermediately resistant (minimum inhibitory concentration = 0.12-1 µg/mL).
- a smaller percentage demonstrate high-level resistance (minimum inhibitory concentration > 2 µg/ml).
- Penicillin or ampicillin remain the treatment of choice for penicillin-sensitive strains.
- High-dose parenteral penicillin, a 3rd-genera-tion cephalosporin or an oral amoxicillin used for intermediate-sensitivity strains, except for meningitis.
- A respiratory fluoroquinolone (gatifloxacin, moxifloxacin, levofloxacin) is used for strains with high-level resistance. Avoid fluoroquinolones in meningitis, and cover with vancomycin.
- Mortality is approximately 5%; prognosis is worse for infants and for patients older than 65 years of age, and for those whose treatment is delayed or who have capsular types 2 or 3, multilobar pneumonia, bacteremia or meningitis, or jaundice, or who are pregnant, have an underlying disease, or alcohol intoxication.
- The 23-valent pneumococcal vaccine is safe and efficacious. It should be given to patients who are over 65 years of age, who have a chronic disease, and who are asplenic, immunocompromised, or alcoholic.
Prevention
Despite the use of antibiotics, mortality during the first 36 hours of hospitalization has not changed. To prevent early mortality and to reduce the incidence of S. pneumoniae infection — the penicillin-sensitive and penicillin-resistant strains alike — vaccination is strongly recommended for all patients with chronic illnesses or those over the age of 65 years.
Generation of specific antibodies directed against the bacterial cell wall confer, prevent, or reduce the severity of disease. Polyvalent vaccine containing antigens to 23 capsular types is available and is effective (approximately 60% reduction of bacteremia in immunocompetent adults). Efficacy decreases with age and is not measurable in immunocompromised patients. The vaccine has proved to be safe and inexpensive, and should be widely used.
Haemophilus influenzae
Group В and non-typable H. influenzae can both cause community-acquired pneumonia. Infection with non-typable H. influenzae is more common in elderly individuals and in smokers with chronic obstructive pulmonary disease. The onset of symptoms tends to be more insidious than that seen with S. pneumoniae, but the clinical pictures are otherwise indistinguishable. A Chest X-Ray can demonstrate lobar or patchy infiltrates, and sputum Gram stain reveals small gram-negative pleomorphic coccobacillary organisms.
Because of their small size and their color, which is similar to background material, H. influenzae may be missed by an inexperienced diagnostician. For the patient requiring hospitalization, intravenous ceftriaxone or cefotaxime is recommended. For oral antibiotic treatment, amoxicillin-clavulanate is effective. However, a number of other oral antibiotics, including trimethoprim-sulfamethoxazole, the newer macrolides (azithromycin and clarithromycin), the fluoroquinolones, and the extended-spectrum cephalosporins (cefpodoxime, cefixime) are also active against this organism.
About Haemophilus influenzae Pneumonia
- This small, gram-negative, pleomorphic coccobacilli is aerobic. It may be mistaken for the background material on sputum Gram stain.
- Non-typable strains are more common in eled-erly people and in smokers with COPD.
- Clinically, Haemophilus influenzae is similar to S. pneumoniae, with a somewhat slower onset.
- Parenteral ceftriaxone or cefotaxime should be used to treat hospitalized patients. Multiple oral regimens-amoxicillin-clavulanate, newer macrolides, fluoroquinolones, and extended-spectrum cephalosporins are useful in outpatients.
Staphylococcus aureus
Fortunately community-acquired pneumonia attributable to S. aureus is rare. The most common predisposing factor is a preceding influenza infection. An increase in the incidence of S. aureus pneumonia is often a marker for the onset of an influenza epidemic. S. aureus pneumonia is also more common in intravenous drug users and in AIDS patients, in association with P. jirovecii pneumonia.
In a few communities, community-acquired methi-cillin-resistant S. aureus (Community-acquired methicillin-resistant Staphylococcus aureus) pneumonia has been described in addition to methicillin-sensitive S. aureus (MSSA). The clinical manifestations of this infection are similar to other forms of bacterial pneumonia. However the illness is often severe, being associated with high fever and a slow response to conventional therapy. A Chest X-Ray can demonstrate patchy infiltrates or dense diffuse opacifications. S. aureus produces multiple proteases that allow this bacterium to readily cross the lung fissures and simultaneously involve multiple lung segments. This broader involvement explains the typical bronchopneumonia pattern on Chest X-Ray. The rapid spread and aggressive destruction of tissue also explains the greater tendency of S. aureus to form lung abscesses and induce a pneu-mothorax. Spread of this infection to the pleural space can result in empyema (seen in 10% of patients). Sputum Gram stain reveals sheets of polymorphonuclear leukocytes and an abundance of gram-positive cocci in clusters and tetrads, and culture readily grows S. aureus. Blood cultures may also be positive.
The treatment of choice for MSSA is high-dose intravenous nafcillin or oxacillin. For methicillin-resistant Staphylococcus aureus pneumonia, vancomycin is generally recommended. The dose of vancomycin should be adjusted to maintain a trough level of 15 to 20 µg/mL to assure therapeutic levels in the lung. Linezolid is an expensive alternative that has equivalent efficacy.
About Staphylococcus aureus Pneumonia
- These large gram-positive aerobic cocci form tetrads and clusters.
- The disease most commonly follows influenza, and is seen in patients with AIDS and in IV drug abusers.
- Destructive bronchopneumonia is complicated by
- lung abscesses,
- pneumothorax,and
- empyema.
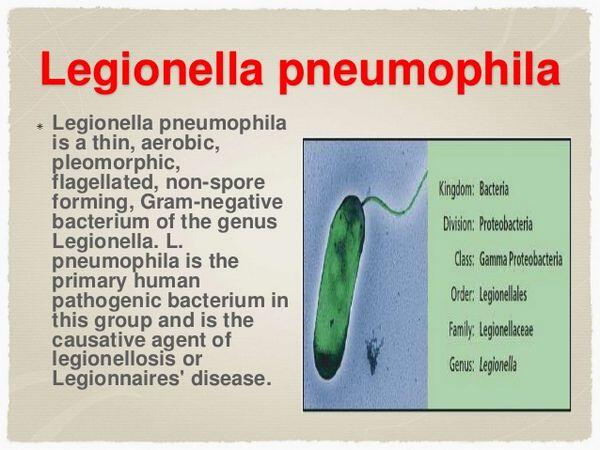
Legionella pneumophila
Legionella species are gram-negative bacilli found throughout the environment in standing water and soil. Infection most commonly results from inhalation of water droplets contaminated with Legionella. Cooling towers or shower heads are most often responsible for aerosolizing contaminated water. Less commonly nosocomial infection has resulted from the use of unsterilized tap water in respiratory therapy devices. Outbreaks of Legionella pneumonia have also been associated with soil excavation. Immunocompromised patients, smokers, and elderly people are more susceptible to this infection. Clinically, Legionella infection causes symptoms typical of other acute community-acquired pneumonias, including high fever, cough, myalgias, and shortness of breath. As compared with other bacterial pneumonias, cough usually produces only small amounts of sputum. Gastrointestinal symptoms, confusion, and headache are more frequently encountered in patients with Legionella. Laboratory findings are similar to other acute pneumonias. The only distinctive finding may be hyponatremia, which is noted in approximately one third of patients. A Chest X-Ray frequently demonstrates lobar pneumonia. In the immunocompromised host, cavitary lesions may be seen. Small pleural effusions are also commonly found.
Diagnosis requires a high index of suspicion, because sputum Gram stain reveals only acute inflammatory cells. The microbiology laboratory must be alerted to the possibility of Legionella species to assure that sputum samples are cultured on buffered-charcoal yeast-extract agar with added suppressive antibiotics. Legionella can also be identified by direct fluorescent antibody staining, although the sensitivity of this technique is low (30% to 50%). Amplification of Legionella Deoxyribonucleic acid from sputum samples by Polymerase chain reaction is available in certain reference laboratories, but not commercially. For L. pneumophila serogroup 1, the most common cause of Legionella pneumonia in the United States (>80% of cases), a highly sensitive and specific urinary antigen test is commercially available. The antigen is excreted early in the illness and persists for several weeks.
For mild disease, an oral macrolide, fluoroquinolone, or tetracycline may used. However, in more severe disease, high doses of intravenous azithromycin or a fluoroquinolone (ciprofloxacin or levofloxacin) are recommended. In transplant patients, a fluoroquinolone is preferred because the macrolides interfere with cyclosporin or tacrolimus metabolism. In the immuno-competent patient, therapy should be continued for 5 to 10 days with azithromycin and for 10 to 14 days with a fluoroquinolone. In the immunocompromised patient, therapy needs to be prolonged for 14 to 21 days to prevent relapse. Mortality is high in legionnaires’ disease, being 16% to 30% in community-acquired disease and up to 50% in hospitalized patients.
About Legionella Pneumonia
- These aerobic gram-negative bacteria do not take up Gram stain well.
- Found in soil and standing water. Aerosolized by cooling towers and shower heads. Also contracted after soil excavation.
- Elderly people, smokers, and immunocompro-mised patients are at increased risk.
- Similar to other acute pneumonias. Somewhat unique characteristics include
- minimal sputum production,
- confusion and headache,
- gastrointestinal symptoms, and
- hyponatremia.
- Diagnostic techniques include
- culture on buffered-charcoal yeast-extract a gar,
- direct fluorescent antibody stain (low sensitivity),
- polymerase chain reaction (still experimental), and
- urinary antigen to serotype I (causes 80% of infections), which is sensitive and specific, and persists for several weeks.
- Azithromycin or a fluoroquinolone are the treatments of choice. In transplant patients, fluoroquinolones are preferred. Mortality is high: 16% to 50%.
Atypical Pneumonia
The atypical forms of pneumonia tend to be subacute in onset, with patients reporting up to 10 days of symptoms before seeking medical attention. Atypical pneumonia is associated with a nonproductive cough, and clinical manifestations tend to be less severe. It is important to keep in mind that significant overlap occurs in the clinical manifestations of this group of infections and the more typical forms of pneumonia associated with purulent sputum production.
Mycoplasma pneumoniae is one of the most frequent causes of “walking pneumonia.” This infection is seen primarily in patients under age 40 years; it is an uncommon cause of pneumonia in elderly individuals. The disease is seasonal, with the highest incidence of Mycoplasma being seen in the late summer and early fall. Sore throat is usually a prominent symptom, and bullous myringitis is seen in 5% of cases. Presence of this abnormality is highly suggestive of Mycoplasma. Tracheobronchitis results in a hacking cough that is often worse at night and that persists for several weeks. Physical exam may reveal some moist rales, but classically, radiologic abnormalities are more extensive than predicted by the exam. Findings on Chest X-Ray consist of unilateral or bilateral patchy lower-lobe infiltrates in a bronchial distribution. The clinical course is usually benign. Fever, malaise, and headache usually resolve over 1 to 2 weeks, but cough can persist for 3 to 4 weeks. Peripheral white blood cell is usually less than 10,000. And sputum Gram stain and culture reveal only normal mouth flora and a moderate inflammatory response.
Diagnosis is made by history and clinical manifestations. Epidemiologic history of contact with a person having similar symptoms is particularly helpful. Currently, no definitive test is available. Sputum Polymerase chain reaction has been found to be sensitive and specific, but that test is not commercially available. Cold agglutinin titers in excess of 1:64 support the diagnosis and correlate with severity of pulmonary symptoms, but are not cost effective. Complement fixation antibody titers begin to rise 7 to 10 days after the onset of symptoms.
Because a reliable, rapid diagnostic test is not currently available, therapy is usually empiric. A macrolide or tetracycline is the treatment of choice; alternatively, a fluoroquinolone can be administered. Azithromycin is the preferred agent when Mycoplasma is suspected, and a standard 5-day course is effective in most cases.
Chlamydia pneumoniae (Taiwan acute respiratory agent) is another important cause of atypical pneumonia. This pathogen is a common cause of community-acquired pneumonia, representing 5% to 15% of cases. The disease occurs sporadically and presents in a manner similar to Mycoplasma, with sore throat, hoarseness, and headache in addition to a nonproductive cough. Radio-logic findings are also similar to those with Mycoplasma. No rapid diagnostic test is widely available, and treatment is empiric. A tetracycline is considered the treatment of choice, but macrolides and fluoroquinolones are also effective.
The final major group of organisms that cause atypical pneumonia is the respiratory viruses: influenza A and B, adenovirus, parainfluenza virus, and respiratory syncytial virus. The respiratory syncytial virus infects primarily young children, elderly people, and the immunocompromised host. These viruses can all present with a nonproductive cough, malaise, and fever.
About Atypical Pneumonia
- These diseases tend to besubacute in onset.
- Cough is nonproductive.
- Illness is often less severe than in other community-acquired pneumonias: “walking pneumonia.”
- Findings in a chest radiograph are usually worse than the physical findings.
- Three primary causes:
- Mycoplasma pneumoniae
- Chlamydophila pneumoniae
- Respiratory viruses: influenza, adenovirus, parainfluenza,and respiratory syncytial virus.
- Rapid tests are available for influenza, but not for Mycoplasma or Chlamydia.
- Treatment with a macrolide or tetracycline is recommended. If influenza is diagnosed, give amantadine, ranitidine, or a neuramidase inhibitor within 48 hours of illness onset.
Auscultatory findings are minimal, and lower lobe infiltrates are generally observed on Chest X-Ray. The clinical virology laboratory can culture each of these viruses from sputum or a nasopharyngeal swab. Rapid commercial tests (10 to 20 minutes) are available for detection of influenza (Quick View, Flu OlA, and Zstatflu). These tests have a sensitivity of 57% to 77%, and all three can distinguish between types A and B.
If influenza A virus is diagnosed, early treatment of the virus with amantadine or rimantadine is recommended. Neuramidase inhibitors are also available, and these agents have activity against both influenza A and B. The influenza vaccine is safe and efficacious, and should be given annually in October through early November to patients over 65 years of age, individuals with serious underlying diseases, nursing home residents, and health care workers.
Aspiration Pneumonia
Case 2
A 35-year-old white man arrived in the emergency room complaining of left-sided chest pain during the preceding 4 days. He had begun drinking large quantities of alcohol 8 days earlier. He vaguely recalled passing out on at least two occasions. He developed a persistent cough, productive of green sputum, 4 days before admission. At that time, he also began experiencing left-sided chest pain on deep inspiration (pleuritic pain). Initially these pains were dull; however, over the next few days, they became increasingly sharp.
Physical exam showed a temperature of38°C and a respiratory rate of 42 per minute. This was a disheveled man, looking older than his stated age, breathing shallowly and rapidly, in obvious pain.
A check of the throat revealed a good gag reflex, extensive dental caries, several loose teeth, severe gingivitis, and foul-smelling breath and sputum. Decreased excursion of the right lung was noted, and the right lower lung field was dull to percussion. Bronchovesicular breath sounds were heard diffusely (inspiratory and expiratory breath sounds of equal duration); moist, medium rales were heard in the right lower and left lower lung fields. Egophony and whispered pectoriloquy were also heard in these areas.
Laboratory workup showed a hematocrit of 50%; a white blood cell count of 21,400/mm3, with 79% polymorphonuclear leukocytes, 7% bands, 1% lymphocytes, and 13% monocytes. Blood gasses showed a pH of 7.46, Pao2 of 56 mm Hg, and a Paco2 of 36 mm Hg. Sputum Gram stain revealed many polymorphonuclear leukocytes and a mixture of gram-positive cocci, gram-positive rods, and gram-negative rods. A Chest X-Ray demonstrated dense right lower lobe infiltrate.
While on antibiotics, this patient continued to complain of chest pain and developed decreased breath sounds in the right lower lobe associated with dullness to percussion. A repeat Chest X-Ray and CTscan demonstrated a large right pleural effusion [see Figure 4-4(A)], and thoracentesis revealed more than 100,000 polymorphonuclear leukocytes/mm3, pleural fluid pH of 7.0, and total protein 3.4 mg/mL Gram stain showed a mixture of gram-positive cocci and gram-positive and gram-negative rods.
Aspiration pneumonia should be suspected in patients with a recent history of depressed consciousness and in patients with a poor gag reflex or an abnormal swallowing reflex. The elderly patient who has suffered a stroke is particularly susceptible to aspiration. In case 2, the patient’s heavy consumption of alcohol led to depression in consciousness.
Three major syndromes are associated with aspiration:
- Chemical burn pneumonitis. Aspiration of the acidic contents of the stomach can lead to a chemical burn of the pulmonary parenchyma. Aspiration of large quantities of fluid can result in the immediate opacification of large volumes of lung. Acid damage causes pulmonary capillaries to leak fluid, release cytokines, and permit infiltration by polymorphonuclear leukocytes. In some patients, noncardiogenic pulmonary edema or adult respiratory distress syndrome develops. Onset of symptoms occurs immediately after aspiration.
- Bronchial obstruction resulting from aspiration of food particles. The inhalation of solid particles results in mechanical obstruction and interferes with ventilation. The patient immediately becomes tachypneic.
- Pneumonia resulting from a mixture of anaerobic and aerobic mouth flora. This form of pneumonia develops several days after aspiration of mouth flora. Patients with severe gingivitis have higher bacterial colony counts in the mouth, and they aspirate a higher inoculum of organisms, increasing the likelihood of a symptomatic pneumonia.
Case 2 had poor dental hygiene and severe gingivitis, predisposing him to the latter form of pneumonia. Often, the sputum is putrid-smelling as a result of the high number of anaerobes. Necrosis of tissue is common in this infection, resulting in the formation of lung abscesses. Infection often spreads to the pleura, resulting in pleuritic chest pain as experienced in case 2. Pleural effusions filled with bacteria and polymorphonuclear leukocytes can develop as observed in this case. Effusions containing bacteria and large numbers of polymorphonuclear leukocytes are called empyemas. Necrosis of the pleural lining and lung parenchyma can result in formation of a fistula tracking from the bronchus to the pleural space. Development of a bronchopleural fistula prolongs hospitalization and may eventually require surgical repair.
Diagnosis
Sputum is often foul-smelling as a result of the high numbers of anaerobic bacteria. Sputum Gram stain reveals many polymorphonuclear leukocytes and a mixture of gram-positive and gram-negative organisms. Sputum culture usually grows normal mouth flora. When aspiration occurs in the hospitalized patient, the mouth often is colonized with more resistant gram-negative organisms plus S. aureus. In these patients, a predominance of gram-negative rods or gram-positive cocci in clusters may be seen on Gram stain, and gram-negative rods or S. aureus may be cultured from the sputum.
A Chest X-Ray reveals infiltrates in the dependent pulmonary segments. When aspiration occurs in the upright position, the lower lobes are usually involved, more commonly the right lower lobe than the left. This difference has an anatomic explanation. The right bronchus divides from the trachea at a straighter angle than does the left main-stem bronchus, increasing the likelihood that aspirated material will flow to the right lung. When aspiration occurs in the recumbent position, the superior segments of the lower lobes or the posterior segments of the upper lobes usually become opacified.
About Aspiration Pneumonia
- Can occur in cases of loss of consciousness, poor gag reflex, or difficulty swallowing.
- Three forms of aspiration:
- Aspiration of gastric contents leads to pulmonary burn and noncardiogenic pulmonary edema.
- Aspiration of an obstructing object causes atelectasis and immediate respiratory distress.
- Aspiration of mouth flora, when associated with poor dental hygiene and mixed mouth aerobes and anaerobes, can lead to foul smelling sputum and eventually lung abscess and empyema. Hospital-acquired aspiration causes gram-negative and Staphy-lococcus aureus pneumonia.
- Treatment depends on the form of the disease:
- Penicillin or clindamycin for community-acquired infection.
- Third-generation cephalosporin and metronidazole for hospital-acquired infection.
- Bronchoscopy for obstructing foreign bodies.
Treatment
Clindamycin or penicillin are both effective antibiotic coverage for community-acquired aspiration pneumonia because they kill both aerobic and anaerobic mouth flora. In cases in which lung abscess has developed, clindamycin has been shown to be slightly superior.
In nosocomial aspiration, broader coverage with a 3rd-generation cephalosporin combined with metronida-zole is generally recommended. Alternatively, a semisyn-thetic penicillin combined with a β-lactamase inhibitor (ticarcillin-clavulanate or piperacillin-tazobactam) or a carbapenem (imipenem or meropenem) can be used.
If aspiration of a foreign body is suspected, bronchoscopy is required to remove the foreign material from the tracheobronchial tree.
Rarer Causes of Community-Acquired Pneumonia
Actinomycosis
Actinomyces species are microaerophilic or anaerobic gram-positive rods that can be part of the polymicrobial flora associated with aspiration pneumonia, particularly in patients with poor oral hygiene. Disease is most commonly caused by Actinomyces israelii.
Actinomycosis pulmonary infection is often indolent and slowly progressive. Lung parenchymal lesions are usually associated with pleural infection, resulting in a thickened pleura and empyema. This organism can break through fascial planes. Spontaneous drainage of an empyema through the chest wall should strongly suggest the possibility of actinomycosis. “Sulfur granules” are often found in purulent exudate; they consist of clusters of branching Actinomyces filaments.
Gram stain reveals branching forms that are weakly gram-positive. These forms can be differentiated from Nocardia by modified stain for acid-fast bacilli, Actinomyces being acid-negative and Nocardia being acid-positive. The organism should be cultured under anaerobic conditions, and grows slowly, with colonies usually requiring a minimum of 5 to 7 days to be identified. Growth can take up to 4 weeks.
High-dose intravenous penicillin (18 to 24X106 U daily) is recommended for 2 to 6 weeks, followed by oral penicillin therapy for 6 to 12 months. Therapy must be continued until all symptoms and signs of active infection have resolved. Other antibiotics that have been successfully used to treat actinomycosis include erythromycin, tetracyclines, and clindamycin.
About Actinomycosis
- These branching gram-positive bacteria are microaerophilic or anaerobic, slow growing, modified acid-fast negative.
- Infection is associated with poor oral hygiene.
- Slowly progressive infection, breaks through fascial planes, causes pleural effusions and fistula tracks, forms “sulfur granules.”
- Alert clinical microbiology to hold anaerobic cultures.
- Treatment must be prolonged: high-dose intravenous penicillin for 2 to б weeks, followed by 6to 12 months of oral penicillin.
Nocardiosis
Nocardia is an aerobic gram-positive filamentous bacterium that often has to be differentiated from Actinomyces. Nocardia is ubiquitous in the environment, growing in soil, organic matter, and water. Pneumonia occurs as a consequence of inhaling soil particles. The number of species causing human disease is large and includes N. abscessus, N. brevicatenalpaucivorans complex, N. nova complex, N. transvalensis complex, N. farcinica, N. asteroides complex, N. brasiliensis, and N. pseudobrasiliensis.
About Nocardiosis
- Nocardia are gram-positive branching bacteria, aerobic, slow growing, modified acid-fast.
- Ubiquitous organism found in the soil.
- Inhalation of soil particles leads to pneumonia.
- The organism infects
- immunocompromised patients (causing disseminated disease in AIDS),
- normal hosts, and
- patients with alveolar proteinosis.
- Pulmonary infection can lead to bacteremia and brain abscess that can mimic metastatic lung carcinoma.
- Alert clinical microbiology to use selective media and to hold cultures.
- Treatment must be prolonged. High-dose parenteral trimethoprim-sulfamethoxazole for at least 6 weeks, followed by oral treatment for б to 12 months.
Infection more commonly develops in patients who are immunocompromised; however, 30% of cases occur in otherwise normal individuals. Patients with AIDS, organ transplant, alcoholism, and diabetes are at increased risk of developing nocardiosis. In addition to pulmonary disease, these patients are at increased risk for developing disseminated infection. Patients with chronic pulmonary disorders, in particular patients with alveolar proteinosis, have an increased incidence of pulmonary Nocardia infection.
Onset of pulmonary disease is highly variable. In some cases, onset is acute; in others, onset is gradual. Symptoms are similar to other forms of pneumonia. A Chest X-Ray may reveal cavitary lesions, single or multiple nodules, a reticular nodular pattern, interstitial pattern, or a diffuse parenchymal infiltrate. Nocardia pulmonary infection often seeds the bloodstream and forms abscesses in the cerebral cortex. The combination of a lung infiltrate with a Central nervous system lesion or lesions is often mistaken for lung carcinoma with Central nervous system metastasis.
Diagnosis is made by sputum examination or lung or cerebral cortex biopsy. Gram stain demonstrates weakly gram-positive branching filamentous forms that are acid-fast on modified acid-fast bacilli stain. On tissue biopsy, organisms are demonstrated on Brown-Brenn or methenamine silver stain. The organism is slow growing and is frequently overgrown by mouth flora on conventional plates. The clinical laboratory should be alerted to the possibility of Nocardia so that they can incubate bacteriologic plates for a prolonged period and use selective media.
Most Nocardia are sensitive to sulfonamides and trimethoprim. Trimethoprim-sulfamethoxazole is generally accepted as the treatment of choice, with a daily dose of 2.5 to 10 mg/kg of the trimethoprim component. High-dose therapy should be continued for at least 6 weeks, followed by lower doses for 6 to 12 months. Some Nocardia species are resistant to sulfonamides, but they are sensitive to amikacin, imipenem, 3rd-generation cephalosporins, minocy-cline, dapsone, and linezolid. Whenever possible, culture and antibiotic sensitivities should be used to guide antibiotic therapy.
Nosocomial (Hospital-Acquired) Pneumonia
Pneumonia is the second most common form of nosocomial infection. It accounts for 13% to 19% of all nosocomial infections. Hospital-acquired pneumonia is defined as a pneumonia that develops 48 hours or longer after hospitalization and that was not developing at the time of admission. Nosocomial pneumonia is a very serious complication and represents the leading infectious-related cause of death in the hospital, the mortality being roughly 1 of every 3 cases. Development of pneumonia in the hospital prolongs hospitalization by more than 1 week.
The condition that most dramatically increases the risk of nosocomial pneumonia is endotracheal intubation. Endotracheal tubes bypass the normal protective mechanisms of the lung, and they increase the risk of pneumonia by a factor of between 6 and 21. It has been estimated that the risk of pneumonia while on a ventilator is 1% to 3% daily. Other factors that increase the risk of pneumonia include age greater than 70 years; Central nervous system dysfunction, particularly coma, leading to an increased likelihood of aspiration; other severe underlying diseases; malnutrition; and metabolic acidosis. Patients on sedatives and narcotics have depressed epiglottal function and are also at increased risk of aspiration. Corticosteroids and other immunosuppressants reduce normal host defenses and allow bacteria to more readily invade the lung parenchyma.
Aerobic gram-negative bacteria account for more than half the cases of nosocomial pneumonia.
About Nosocomial Pneumonia
- Pneumonia is one of the most common nosocomial infections.
- Risk factors include
- endotracheal intubation (20 times the baseline risk, 1% to 3% incidence daily),
- age greater than 70 years,
- depressed mental status,
- underlying disease and malnutrition, and
- metabolic acidosis.
- Primary causes are gram-negative bacilli and Staphylococcus aureus.
- Colonization is difficult to differentiate from infection. Bronchoscopy is not helpful. Factors that favor infection include:
- worsening fever and leukocytosis with left shift;
- sputum Gram stain with increased polymorphonuclear leukocytes, predominance of one organism;
- decreasing Pao2 indicative of pulmonary shunting; and
- expanding infiltrate on chest radiographs.
- Broad-spectrum empiric therapy can be initiated after samples are obtained for culture, but coverage should be adjusted based on culture results and clinical response.
Escherichia coli, Klebsiella, Serratia, Enterobacter, and Pseudomonas species represent the most common gram-negative rods. S. aureus is the most common gram-positive pathogen, causing 13% to 40% of nosocomial pneumonias. The risk of S. aureus infection is higher in patients with wound infections or burns, and it is also higher in intubated patients with head trauma or neurosurgical wounds. Anaerobes are often isolated in nosocomial pneumonia, but they are thought to be the primary agent in only 5% of cases. S. pneumoniae is seldom the cause of pneumonia in the patient who has been hospitalized for more than 4 days.
Diagnosis of true pneumonia is often difficult in the intubated patient. In elderly patients with chronic bronchitis and congestive heart failure or adult respiratory distress syndrome, definitively proving that the patient has or does not have an infection is often impossible. Differentiating infection from colonization represents a critical branch point in the appropriate management of antibiotics. Within 3 to 5 days of antibiotic initiation, the mouth flora and the flora colonizing the tracheo-bronchial tree change. A change in the organisms growing from sputum culture is therefore to be expected and does not in itself indicate that the patient has a new infection. The change simply documents colonized of the patient with resistant flora. For example, in a high percentage of patients receiving broad-spectrum antibiotics, Candida albicans begins to grow in sputum cultures because of the reduction in the competing bacterial mouth flora. However, that organism does not invade the lung and almost never causes airborne pneumonia. Antifungal coverage is therefore not required unless the patient develops symptomatic thrush.
Evidence supporting the onset of a new infection includes
- a new fever or a change in fever pattern;
- a rise in the peripheral white blood cell count, with a increase in the percentage of polymorphonuclear leukocytes and band forms (left shift);
- Gram stain demonstrating increased number of polymorphonuclear leukocytes in association with a predominance of bacteria that are morphologically consistent with the culture results;
- increased purulent sputum production from the endotracheal tube;
- reduced arterial PaO2, indicating interference with alveolar-capillary oxygen exchange; and
- enlarging infiltrate on Chest X-Ray.
Multiple studies have used bronchoscopy with protected brushings or bronchial lavage and quantitative cultures and Gram stains. A randomized trial found that that samples obtained by bronchoscopy provide no advantage over endotracheal suction, and therefore that procedure is not recommended in ventilation-associated pneumonia.
When infection is likely or the patient is extremely ill, and when a new pulmonary infection cannot be convincingly ruled out, antibiotics should be quickly started; or, if the patient is receiving antibiotics, the regimen should be changed to cover for antibiotic-resistant bacteria. In the absence of specific findings indicative of infection, colonization is more likely, and the antibiotic regimen should not be changed.
Indiscriminate modifications of antibiotic therapy eventually select for highly resistant pathogens that are difficult — or in some cases impossible — to treat. Switches to broader-spectrum, more powerful antibiotics should be undertaken cautiously, and should be initiated only when convincing evidence for a new infection is present. In the patient who is deteriorating clinically, broader-spectrum coverage can be temporarily instituted once blood, urine, and sputum samples for culture and Gram stain have been obtained. The 3-day rule should then be applied, with the antibiotic regimen being modified within 3 days, based on the culture results, so as to prevent colonization with even more highly resistant bacteria.
These regimens are recommended for nosocomial pneumonia:
- Third-generation cephalosporin (ceftriaxone, cefotaxime, ceftizoxime, or ceftazidime)
- Cefepime
- Ticarcillin-clavulanate or piperacillin-tazobactam
- Imipenem or meropenem
An aminoglycoside (gentamicin, tobramycin, or amikacin) may or may not be added. If P. aeruginosa is suspected, ciprofloxacin, piperacillin-tazobactam, ticarcillin-clavulanate, cefepime, aztreonam, imipenem, or meropenem should be used. Many experts recommend administration of two agents from different classes to prevent development of resistance. Aminoglycosides should never be used alone to treat Pseud, aeruginosa because the antibiotic levels achievable in the lung are low. Aerosolized tobramycin (80 mg twice daily) has proven to be useful adjunctive therapy. If Staph. aureus is suspected, van-comycin should be added pending culture and sensitivity results. Specific anaerobic coverage is usually not required in the absence of clear aspiration.
Empyema
Causation
Infection of the pleural space is most commonly the consequence of spread of pneumonia to the parietal pleura. More than half of empyema cases are associated with pneumonia. The most common pathogens in this setting are S. pneumoniae, S. aureus, S. pyogenes, and anaerobic mouth flora. Empyema is also a complication of trauma and surgery, and when those are the inciting factors, S. aureus and aerobic gram-negative bacilli predominate. In the immunocompromised patient, fungi and gram-negative bacilli are most commonly encountered.
About Empyema
- Suspect empyema if fever persists despite appropriate antibiotic treatment of pneumonia.
- The condition is most common with Streptococcus pneumoniae, Staphylodcoccus aureus, S.pyogenes,and mouth anaerobes.
- A chest radiograph with lateral decubitus is sensitive; computed tomography scan is also helpful.
- If empyema is being considered, an ultrasound guided thoracentesis should be performed.
- When pH is less than 7.2, glucose is less than 40 mg/dL, and lactate dehydrogenase exceeds 1000 IU/L, empyema is strongly suggested.
- Use tube drainage initially; if loculation continues, urokinase can be given.May require surgical intervention.
- Early diagnosis and drainage prevents lung and pleura compromise.
- Mortality associated with empyema is high: 8% to 15% in young patients, and 40% to 70% in elderly ones.
Pathophysiology
Pleural effusions occur in approximately half of all pneumonias; however, only 5% of pneumonias develop true empyema. Because pleural fluid is deficient in the opsonins, immunoglobulin G, and complement, bacteria that find a way to this culture medium are only ineffectively phagocytosed by polymorphonuclear leukocytes. As polymorphonuclear leukocytes break down in the closed space, they release lysozyme, bacterial permeability-increasing protein, and cationic proteins. These products slow the growth of bacteria, lengthening doubling times by a factor of 20 to 70. The slow growth of the bacteria renders them less sensitive to the cidal effects of antibiotics. In the empyema cavity, pH is low, impairing white blood cell function and inactivating some antibiotics — in particular, the aminoglycosides.
Clinical Manifestations
Persistent fever despite appropriate antibiotic treatment for pneumonia should always raise the possibility of an enclosed pleural infection. Fever is often accompanied by chills and night sweats. Pleuritic chest pain is a common complaint, as is shortness of breath. Physical exam is help-fill in detecting large effusions. As noted in case 4.2, the area in which fluid is collecting is dull to percussion, and breath sounds are decreased. At the margin between fluid and aerated lung, egophony and bronchial breath sounds are commonly heard, reflecting areas of pulmonary consolidation or atelectasis.
On Chest X-Ray, fluid collections as small as 25 mL can alter the appearance of the hemidiaphragm on posterior-anterior view, and on lateral views, 200 mL of fluid is generally required to blunt the posterior costophrenic angle. A lateral decubitus view with the pleural effusion side down can demonstrate layering of 5 to 10 mL of free fluid. Contrast-enhanced chest computed tomography is particularly helpful in differentiating lung abscess from empyema, and it demonstrates the full extent of the effusion and the degree of pleural thickening.
Ultrasound is very useful in determining the dimensions of the effusion, and it is the most effective method for guiding thoracentesis. Septations are readily visualized by this technique and indicate the development of a loculated collection that requires drainage. Ultrasound guidance of thoracentesis is strongly recommended because of the associated decreased incidence of complicating pneumothorax. The fluid should be analyzed for cellular content, and Gram stain, fungal stain, acid-fast bacilli stain, and aerobic and anaerobic cultures should be obtained. If the fluid is frankly purulent, the pleural space should be completely drained. If the fluid is not overtly purulent, the fluid should also be analyzed for pH, glucose, lactate dehydrogenase, and total protein. A pleural fluid pH below 7.2, a glucose level below 40 mg/dL, and a lactate dehydrogenase level above 1000 IU/L are consistent with empyema and justify pleural fluid drainage to prevent loculation, pleural scarring, and restrictive lung disease.
Treatment
Antibiotic therapy for the offending pathogen is of primary importance, and antibiotic coverage depends on the pathogen identified by sputum or pleural fluid Gram stain and culture. When a significant pleural fluid collection is apparent, a more prolonged course of antibiotics (2 to 4 weeks) is generally required.
Parapneumonic effusions that move freely and that are less than 1 cm in width on lateral decubitus film can be managed medically; thoracentesis is not required. If the collection is larger or does not flow freely, thoracentesis should be performed. If biochemical evidence for empyema is present, drainage by chest tube is recommended. Repeated thoracentesis is rarely successful in completely draining the pleural fluid collection unless the fluid has a thin viscosity and is present in small volumes. Drainage by closed chest tube is usually successful with smaller effusions occupying up to 20% of the hemithorax, but it is often ineffective when the volume of fluid occupies more than 40% of the hemithorax. Interventional radiology is required to precisely place French catheters at sites of loculation and to break up areas of adhesion under computed tomography guidance. If tube drainage proves ineffective after 24 hours, intrathoracic urokinase (125,000 U diluted in 50 to 100 mL sterile normal saline) should be instilled to break down intrapleural fibrin and encourage free drainage of infected fluid. If thoracentesis and urokinase are unsuccessful, operative intervention is required.
Empyema is a serious complication, with an associated 8% to 15% mortality in young, previously healthy patients and 40% to 70% mortality in patients who are elderly or have significant underlying disease. Patients with nosocomial pathogens and polymicrobial infection also have a worse prognosis. Delay in diagnosis and appropriate drainage increases the need for surgical resection of the pleura and manual re-expansion of the lung.







