Essentials of Diagnosis
- Transmission in crowded or closed populations.
- Meningitis most common, but bacteremia, pneumonia, and other syndromes can occur in absence of meningitis.
- Disease often associated with petecchial or purpuric rash, which may involve palms and soles.
- Neutrophil-predominant cell profile in CSF, with low-glucose and high-protein concentrations.
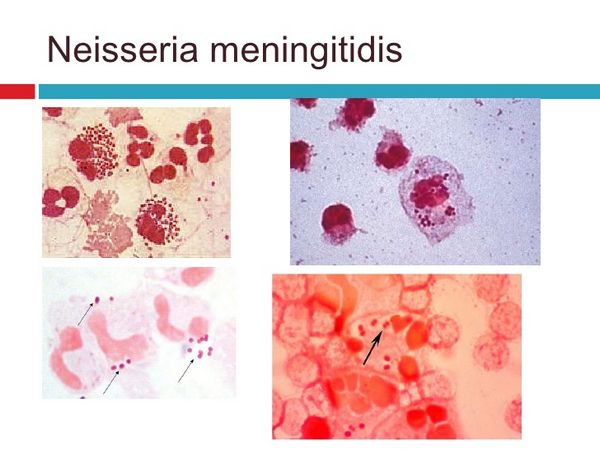
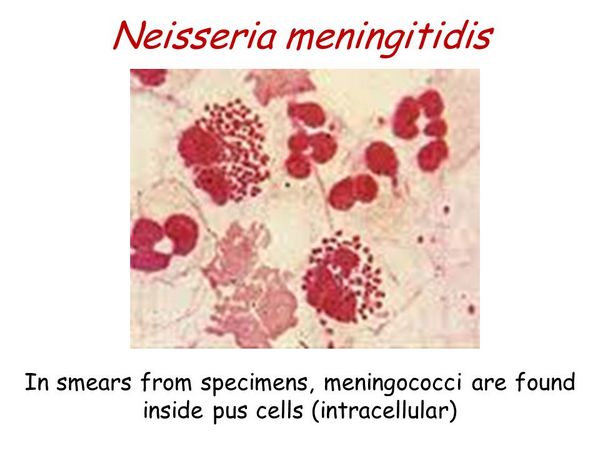
- Gram-negative diplococci inside neutrophils on stained smears of CSF and aspirated skin lesions.
- Fastidious organism requires special media and growth conditions.
- Blood and CSF most useful specimens for recovery of organism.
- Improved sensitivity of detection with PCR in CSF, but not widely available.
General Considerations
Epidemiology
N meningitidis is found only in humans and is a member of the normal oropharyngeal flora in 5-15% of healthy adults and children. In crowded or closed populations such as in boarding schools or military camps, higher carriage rates are observed. Transmission is from person to person by the respiratory route through nasopharyngeal secretions. N meningitidis is communicable and spreads within families and communities; there is a 500- to 1200-fold increased risk of developing meningococcal disease among household contacts of an index case in the 30-60 d after exposure, compared with the risk of developing disease among the general population.
Meningococcal disease usually occurs as isolated cases, but sporadic small epidemics occasionally take place. Group A strains in particular tend to cause widespread epidemics in 8- to 12-y cycles, especially in sub-Saharan Africa. The case rates in endemic areas of Africa vary widely, but overall the rate is 10-25 cases per 100,000 population per year. In the United States, the average annual incidence is 1-2 cases per 100,000 population per year. Most cases are seen in children and young adults (ages 5-19 y).
Microbiologic characterization of N meningitidis has been useful in understanding the epidemiology of this disease. The N meningitidis polysaccharide capsule provides the basis for a serogroup typing system. The most important serogroups causing invasive disease are A, B, C, W-135, and Y. These different polysaccharide capsular types are associated with characteristic epidemiological patterns. Group A strains are linked to worldwide epidemics, whereas B and C strains are considered endemic, causing sporadic cases and limited outbreaks. Currently ~ 50% of all cases in the United States are caused by serogroup B and another 20% by serogroup C. The remainder are primarily serogroups Y and W-135. The proportions of disease caused by serogroups C and Y have been rising in recent years. Each serogroup has been further subdivided into serotypes. For example, N meningitidis group B has 12 serotypes based on unique OMPs. These further subtypings of serogroups have been important both for epidemiologic studies and for vaccine development.
Microbiology
See discussion in N gonorrhoeae section.
Pathogenesis
Adherence, colonization, and invasion are the three essential properties of N meningitidis that enable infection in the human host. Before attachment can occur, secretory immunoglobulin A (IgA) must be inactivated. This is accomplished by secretion of a protease that cleaves IgA1. N meningitidis cells attach selectively to nonciliated pharyngeal mucosal cells, leading to colonization of the nasopharynx and upper respiratory tract. The PilC molecule and the OMPs Opa and Opc (opacity proteins) are important for adherence to the nasopharyngeal epithelial cells. The expression of Opa also mediates host cell invasion. The capsular polysaccharide facilitates invasion into the bloodstream and is antiphagocytic. The Opa and Opc groups of proteins have a high variability in their surface expression; this involves variation in promoter activity and frameshifts, which result in reversible on-off switching at the transcriptional and translational levels.
Host genetic factors, as well as other cofactors such as passive smoke and concurrent infection in the upper respiratory tract, appear to play a role in the development of meningococcal disease. Viral infection may increase the development of nasopharyngeal infection with N meningitidis, and thus lead to higher carriage rates in households or groups of exposed individuals. It should be noted however that exposure to pathogenic strains of N meningitidis most often leads to a carrier state, but not always to active infection.
The protection provided by the host complement system is demonstrated by the enhanced susceptibility to bacteremic Neisseria disease of those patients who are complement deficient. The importance of another protective factor, specific IgG antibody to N meningitidis, is demonstrated by the increased incidence of serious N meningitidis infection in children between the ages of 6 mo and 2 y. This time interval corresponds to the time between loss of maternal antibodies and the development of natural immunity. Infections in outbreak situations, eg, in young adults, generally represent new infection with a virulent strain in individuals without specific antibodies.
Meningococcal endotoxin, primarily LOS, induces meningococcal sepsis. LOS mediates production of cytokines such as tumor necrosis factor-alpha (TNF-alpha), interleukin (IL)-1, IL-6, and interferon-gamma. The expression of these host inflammatory mediators results in increased endothelial permeability and myocardial depression and hypotension. Damage to the vascular endothelium results in petecchial or purpuric skin lesions. These lesions reflect a similar process that occurs in multiple organs, leading to shock. Other inflammatory mediators such as prostaglandins, leukotrienes, and platelet-activating factor enhance granulocytic function and intravascular clotting and thrombosis. This in turn leads to disseminated intravascular coagulation, adrenal hemorrhage, decreased vascular resistance, circulatory collapse, and finally death.
Clinical Findings
N meningitidis causes acute bacterial meningitis with or without meningococcemia. Its potential to progress rapidly mandates early recognition and use of empiric antimicrobial treatment. Pharyngitis precedes meningitis in most patients and is followed by fever with chills and malaise. The classic symptoms of meningitis are headache, nausea and vomiting, and stiff neck. A prominent feature is a petecchial rash that starts distally on the extremities and progresses to the trunk. It often involves the palms and soles. Skin lesions evolve over hours and may continue to develop for a day or more despite appropriate antibiotic treatment. Fulminant meningococcemia (Waterhouse-Friderichsen syndrome) occurs in 5-15% of cases. Fulminant disease is associated with vascular collapse, often presaged by apprehension, restlessness, and mental status changes, all developing within a few hours. Acute meningococcemia can occur in the absence of meningitis and presents as the sepsis syndrome.
High fever is usually observed; absence of fever is a poor prognostic sign. Pneumonia from N meningitidis is reported in as many as 15-20% of patients with meningococcemia or meningitis and may occur by itself. Case fatality rates vary according to the prevalence of the disease and socioeconomic conditions. The fatality rate in meningococcal meningitis may be as low as 7% in industrialized countries and as high as 70% for meningococcemia in some developing countries.
Chronic meningococcemia is less common than other bacteremic meningococcal syndromes and presents with fever, headache, rash (90%), and joint pains (66%) that occur intermittently or over a period of days or weeks. The skin lesions occur with the febrile episodes. These lesions are maculopapular (47%) or petecchial (12%) or erythematous and nodular with a pustular center (13%). Without diagnosis and treatment, localized infection such as meningitis, endocarditis, or arthritis develops. Chronic meningococcemia is very responsive to antibiotic therapy.
Diagnosis
The diagnosis of meningococcal infection is made by isolation of the organism or detection of its antigen from blood or cerebrospinal fluid (CSF). If possible, blood, CSF, throat swab, and petecchial lesions should be cultured prior to antibiotic use. Collection of these specimens, however, should never delay treatment. Leukocytosis is often as high as 20,000-30,000 cells/mm3. Blood cultures are positive in 50% of cases of meningitis before antibiotics and in ~ 5% of cases after antibiotics are given. Gram stains of skin lesions are positive in ~ 70% of cases. This is a useful diagnostic test especially if antibiotics have been given before the culture specimens could be obtained.
The CSF pleocytosis of meningococcal meningitis is typical of most acute bacterial meningitides with a predominance of PMNs. The CSF glucose is low with a normal or high CSF protein. Gram stain of the CSF is positive in ~ 75% of cases. CSF should be inoculated onto chocolate agar as soon as possible (within minutes) because of the fastidious nature of this organism, like N gonorrhoeae. A latex agglutination test is available and may be indicated for diagnosis in patients treated with antibiotics. This test is expensive and may not improve diagnostic specificity compared with a Gram stain performed by an experienced microscopist. Positive throat and nasopharyngeal cultures in the setting of meningitis should be interpreted with caution, because there are many coincidental meningococcal carriers.
Differential Diagnosis
The differential diagnosis is broad, given the clinical presentation of infections caused by N meningitidis. The primary considerations in the setting of meningitis with a rash should include the rickettsial diseases such as Rocky Mountain spotted fever, epidemic typhus, and Brill-Zinsser disease. Toxic shock syndrome caused by Staphylococcus aureus may mimic meningococcemia, and therefore additional appropriate antimicrobial coverage is required. Other diseases to consider include viral exanthems such as infection with echovirus type 9, typhoid fever, and vasculitis syndromes such as polyarteritis nodosa, Churg-Strauss syndrome, and Henoch-Schonlein purpura.
Treatment
Treatment should be initiated as soon as the diagnosis is considered (Box 3). Intravenous penicillin G or ceftriaxone is the standard of therapy. The duration of therapy is dependent on the clinical response, but 7 d is considered adequate for both meningitis and chronic meningococcemia.
The clinical importance of isolates found to have altered penicillin-binding proteins with intermediate resistance to penicillin (MIC of 0.1-1.0 ug/ml) is unclear, as patients with these strains respond well to penicillin. For patients who do not respond adequately to therapy, the bacterial isolate should be tested for antibiotic resistance, and the therapy changed to ceftriaxone or cefotaxime if the isolate is resistant to penicillin.
N meningitidis has traditionally been exquisitely sensitive to penicillin, but recent reports from Spain, England, South Africa, Argentina, Canada, and the United States indicate an increasing percentage of penicillin-resistant strains. A third-generation cephalosporin such as ceftriaxone should be used if there is resistance in the area or if the microbiologic diagnosis might include Haemophilus influenzae or Streptococcus pneumoniae.
Chloramphenicol is effective for meningococcal infection, although there are reports of resistance in parts of sub-Saharan Africa. Although it is usually considered bacteriostatic, chloramphenicol is bactericidal against the meningococcus and achieves high CSF levels. It remains an excellent alternative in beta-lactam-allergic patients and in situations in which multiple dosing of parenteral drug is not feasible. Data to support the use of corticosteroids as adjunctive therapy in meningococcal meningitis are limited.
Prevention & Control
Antimicrobial chemoprophylaxis should be given to close contacts of patients with meningococcal disease, because this is a primary means of prevention of disease (Box 4). Close contacts are defined as household members, day care center contacts, and anyone directly exposed to the patient’s oral secretions. Antimicrobial prophylaxis should be administered as soon as possible (ideally within 24 h) after case identification, because the secondary attack rate is highest within the first few days of onset of disease in the primary patient. Chemoprophylaxis given > 14 d after the onset of illness in the primary (index) case is of limited or no value. Nasal and oropharyngeal cultures are of no value in determining the need for chemoprophylaxis.
The three antibiotics used for chemoprophylaxis against meningococcal disease are rifampin, ciprofloxacin, and ceftriaxone. Rifampin is the best studied and may be the most efficacious, but it is not the most convenient. It requires multiple dosing, is not recommended in pregnancy, and may cause gastrointestinal side effects. Ciprofloxacin is given as a single dose and is generally well tolerated, but it is not recommended in pregnancy or for children < 18 y of age. Ceftriaxone is also given as a one-time dose but requires intramuscular injection.
A quadrivalent vaccine for meningococcal serogroups A, C, Y, and W-135 is available in the United States. Efficacy varies by age and serogroup. Protection against disease caused by serogroups A and C is 85-100% in older children and adults. The vaccine polysaccharides elicit bactericidal antibody that is serogroup specific. The serogroup B polysaccharide is poorly immunogenic in humans, and thus no useful vaccine against serogroup B is currently available; however, with the recent determination of the complete serogroup B meningococcus genome sequence, a set of immunogenic outer surface proteins from this organism has been revealed. A candidate vaccine has been created from some of these proteins. Serogroup B is the most common cause of meningococcal disease in Europe, North America, and several countries in Latin America.
Vaccine protection decreases over time and more rapidly in young children. In children older than 4 y, one study documented an efficacy of 67% at 3 y after vaccination. The current vaccine is useful in controlling serogroup C meningococcal outbreaks. It is also recommended in the following high-risk groups: (1) complement-deficient hosts (C3, C5-C9), (2) asplenic individuals, (3) travelers to endemic areas, (4) research or laboratory personnel, and (5) military recruits.
BOX 1. Therapy for Neisseria gonorrhoeae Infection
Adults
Children
First choice:
Uncomplicated urethritis, cervicitis, pharyngitis, proctitis, prostatitis, ophthalmia neonatorum, conjunctivitis1
- Ceftriaxone, 125 mg IM, single dose
OR
- Cefixime, 400 mg PO in a single dose
OR
- Ciprofloxacin, 500 mg PO in a single dose
PLUS
- Doxycycline,2 100 mg PO twice per day × 7 d
OR
- Azithromicin, 1 g PO, single dose
- Ceftriaxone, 25-50 mg/kg IV or IM, single dose (not to exceed 125 mg)
OR
- Cefotaxime, 100 mg/kg IV or IM, single dose (not to exceed 1 g)
Alternatives3
- Spectinomycin, 2 g IM, single dose
OR
- Ofloxacin, 400 mg PO, single dose
PLUS
- Doxycycline, 100 mg PO twice per day × 7 d
OR
- Erythromycin base, 500 mg PO four times daily × 7 d
OR
- Amoxicillin, 500 mg PO 3 times daily
- Spectinomycin, 40 mg/kg (not to exceed 2 g) IM, single dose
PLUS
- Erythromycin (base or stearate), 20-40 mg/kg/d in 4 divided doses (not to exceed 2 g/d)
Complicated infections:
Disseminated gonococcal infection (dermatitis-arthritis syndrome), meningitis,4 endocarditis
- Ceftriaxone, 1-2 g IV or IM every 12-24 h for 1-2 d after improvement or 7 d
OR
- Cefotaxime, 1-2 g IV every 8 h for 1-2 d after improvement or 7 d
PLUS
- Doxycycline, 100 mg PO twice daily × 7 d
- Ceftriaxone, 50 mg/kg/d (not to exceed 1 g/d) IV or IM × 7 d
PLUS
- Erythromycin (base or stearate), 20-40 mg/kg/d in 4 divided doses (not to exceed 2 g/d)
Alternatives
- Spectinomycin, 2 g IM every 12 h
OR
- Chloramphenicol, 1 g every 6 h
- Spectinomycin, 40 mg/kg (not to exceed 2 g) IV or IM × 7 d
PLUS
- Erythromycin (base or stearate), 20-40 mg/kg/d in 4 divided doses (not to exceed 2 g/d)
- Irrigate eyes immediately and frequently with saline until discharge is eliminated.
- Doxycycline or tetracycline is given for possible chlamydial co-infection.
- In pregnancy, ceftriaxone and erythromycin should be used; see text.
- Longer duration and higher doses are indicated; see text.
BOX 2. Prevention and Control of Neisseria gonorrhoeae Infection
Prophylactic measures
Avoid sexual exposure: abstinence, barrier methods (ie, condoms).
Chemoprophylaxis (ie, post exposure for sexual assault1)
- Ceftriaxone, 125 mg IM single dose
PLUS
- Metronidazole, 2 g orally in a single dose
PLUS
- Azithromycin, 1 g orally in a single dose
OR
- Doxycycline, 100 mg orally twice a day for 7 d
- Postexposure hepatitis B vaccination (without HBIG)
Control
Case finding by active surveillance.
Treat contacts of known cases (“epidemiologic treatment”) if the risk of unnecessary therapy is less than the risk of developing complications of the infection or the probability of transmission to other contacts
Isolation Precautions
- None
1) These antibiotics treat the likely infections in victims of sexual assault (see N Engl J Med 1990; 322:713).
BOX 3. Therapy for Neisseria meningitidis Infection
Adults
Children
First Choice
- Penicillin G, 24 million U IV per 24 h in divided doses every 2-4 h
- Penicillin G, 0.25 million U/kg/d IV (every 2-4 h for 4-10 d)
Alternatives
- Ceftriaxone, 2 g IV every 12 h
OR
- Chloramphenicol, 50-100 mg/kg/d PO or IV every 6 h
- Ceftriaxone, 80-100 mg/kg/d IV (every 12 h for 4-10 d)
OR
- Chloramphenicol, 50-100 mg/kg/d PO or IV (every 6 h)
BOX 4. Prevention and Control of Neisseria meningitidis Infection
Prophylactic Measures
- Quadrivalent vaccine1 (A, C, Y, W-135) (Menomune) 0.5 ml SC × 1 (may need to be repeated if < 4 y old)
Control with chemoprophylaxis
- Rifampin, 10 mg/kg (= 600 mg) PO every 12 h × 4 doses
- Ciprofloxacin, 500 or 750 mg PO per single dose
- Ceftriaxone, 250 mg IM per single dose (adults); 125 mg IM per single dose (children < 12 y)
Isolation Precautions
- Should remain in respiratory isolation until appropriate antibiotic therapy has been given for 24 h
This vaccine is suggested for high-risk groups (see text).




