Clofazimine
Clofazimine is a phenazine dye with antimycobacterial and anti-inflammatory activity.
Administration
Clofazimine is administered orally. Clofazimine should be taken with a meal to maximize absorption of the drug.
Dosage
Clofazimine dosages exceeding 100 mg daily should be given for as short a period as possible and only under close medical supervision.
Leprosy
The usual adult dosage of clofazimine for the treatment of leprosy is 50-100 mg once daily. Clofazimine also is given in a dosage regimen that includes 50 mg once daily plus an additional 300-mg dose given once monthly.
Multibacillary Leprosy
For the treatment of multibacillary leprosy, the World Health Organization (WHO) and other clinicians recommend that adults receive clofazimine in a dosage of 50 mg once daily (with an additional 300-mg dose given once monthly) in conjunction with rifampin (600 mg once daily) and dapsone (100 mg daily) given for 12 months. Children 10-14 years of age with multibacillary leprosy may receive clofazimine in a dosage of 50 mg once every second day (with an additional 150-mg dose given once monthly) in conjunction with rifampin (450 mg once monthly) and dapsone (50 mg once daily) given for 12 months, and children younger than 10 years of age should receive an appropriately adjusted dosage (e.g., rifampin 300 mg once monthly in conjunction with clofazimine 50 mg twice each week plus 100 mg once monthly and dapsone 25 mg daily) given for 12 months. The WHO recommends supervised administration of some drug doses included in the regimen (e.g., once-monthly doses).

While the 12-month regimen is adequate for most patients with multibacillary leprosy, the WHO recommends that multibacillary leprosy patients with a high bacteriologic index who demonstrate no improvement (with evidence of worsening) of leprosy following completion of the initial 12 months of treatment should receive an additional 12 months of therapy.
If a patient with multibacillary leprosy experiences severe adverse effects related to dapsone, dapsone may be discontinued and therapy continued with rifampin and clofazimine given in the usually recommended dosages. If an adult with multibacillary leprosy will not accept or cannot tolerate clofazimine, the WHO states that these adults may receive supervised administration of a once-monthly rifampin-based multiple-drug regimen (ROM) that includes rifampin (600 mg once monthly), ofloxacin (400 mg once monthly), and minocycline (100 mg once monthly) given for 24 months.
For the treatment of multibacillary leprosy in adults who cannot receive rifampin because of adverse effects, intercurrent disease, or infection with rifampin-resistant Mycobacterium leprae, the WHO recommends supervised administration of a regimen of clofazimine (50 mg once daily), ofloxacin (400 mg once daily), and minocycline (100 mg once daily) given for 6 months, followed by a regimen of clofazimine (50 mg once daily) and either ofloxacin (400 mg once daily) or minocycline (100 mg once daily) given for at least an additional 18 months.
Paucibacillary Leprosy
If a patient with paucibacillary leprosy receiving the usually recommended regimen of rifampin and dapsone experiences severe adverse effects related to dapsone, dapsone may be discontinued from the regimen and clofazimine substituted (using the dosage recommended for the treatment of multibacillary leprosy) for 6 months.
Erythema Nodosum
Leprosum The treatment of erythema nodosum leprosum (ENL) reactions (type 2 reactions) depends on the severity of symptoms. When prolonged corticosteroid therapy is necessary in patients with ENL, clofazimine in a dosage of 100-300 mg daily given in 2 or 3 divided doses for up to 3 months or longer may eliminate or reduce corticosteroid requirements. In patients with severe, corticosteroid-dependent ENL, adequate control (reduction of severity and frequency of attacks and corticosteroid use) may require prolonged initial clofazimine therapy (for up to about 7 months), and extended therapy with the drug (an additional 9-24 months) may be necessary to prevent recurrence.
An initial exacerbation may occur in some patients during the first several weeks of clofazimine therapy, but this generally is followed by progressive improvement.
Clofazimine dosages up to 400 mg daily have been used to control ENL in some adults; however, the manufacturer states that dosages greater than 200 mg daily are not recommended.
Dosage of clofazimine should be reduced to the lowest effective level (e.g., to 100 mg daily) as soon as possible after the reactive episode is controlled. The WHO states that high-dose clofazimine therapy should not exceed 12 months.
Mycobacterium avium
Complex (MAC) Infections When used in the treatment of Mycobacterium avium complex (MAC) infections in adults, clofazimine has been given in a dosage of 100 mg one or 3 times daily in conjunction with other antimycobacterial agents.A clofazimine dosage of 1-2 mg/kg daily has been used for the treatment of MAC infections in children. However, higher dosages (up to 50 mg daily in children younger than 4 years of age, or approximately 4 mg/kg daily) also have been used in children.
Drug Interactions
Dapsone
Results of several studies indicate that concomitant clofazimine does not affect the pharmacokinetics of dapsone, although a transient increase in urinary excretion of dapsone reportedly occurred in a few patients receiving concomitant therapy with the drugs. In a study in lepromatous leprosy patients receiving dapsone (100 mg daily) and rifampin (600 mg daily), concomitant administration of clofazimine (100 mg daily) did not affect plasma dapsone concentrations or the plasma half-life or urinary elimination of dapsone.
There is some evidence that dapsone may decrease or nullify some of the anti-inflammatory effects of clofazimine. In vitro, clofazimine and dapsone have opposing effects on neutrophil motility and lymphocyte transformation. Some clinicians suggest that this theoretically could adversely affect the efficacy of clofazimine in patients with erythema nodosum leprosum (ENL) reactions.Several borderline leprosy and lepromatous leprosy patients with severe, recurrent ENL reactions reportedly required higher clofazimine dosage to control these reactions when dapsone therapy was given concomitantly than when clofazimine was given alone.
The manufacturer of clofazimine, however, suggests that further study is needed to confirm this interaction and states that it is advisable to continue treatment with both clofazimine and dapsone in patients who develop leprosy-associated inflammatory reactions, including ENL, during concomitant therapy with the drugs. There is no evidence to date that clofazimine and dapsone interfere with the antimycobacterial activity of each other.
Rifampin
Concomitant administration of clofazimine in leprosy patients receiving rifampin alone or in conjunction with dapsone reportedly may decrease the rate of absorption of rifampin, delay the time to reach peak serum rifampin concentrations, and result in a slight decrease in the area under the plasma concentration-time curve (AUC) of the drug. However, in a study in lepromatous leprosy patients receiving dapsone (100 mg daily) and rifampin (600 mg daily), concomitant administration of clofazimine (100 mg daily) did not affect plasma rifampin concentrations or the AUC, plasma half-life, or urinary elimination of rifampin.
Isoniazid
In a study in lepromatous leprosy patients receiving clofazimine (300 mg daily), concomitant administration of isoniazid (300 mg daily) apparently resulted in increased plasma and urinary concentrations of clofazimine and decreased concentrations of the drug in skin.
Acute Toxcicity
There currently is no information available on overdosage of clofazimine in humans. The oral LD50 of the drug is 3.3 g/kg in rabbits and greater than 5 g/kg in mice, rats, and guinea pigs. If acute overdosage of clofazimine occurs, the stomach should be emptied by inducing emesis or by gastric lavage, and supportive and symptomatic treatment should be initiated.
Mechanism of Action
Antimycobacterial Effects
The mechanism of action of clofazimine against mycobacteria has not been fully elucidated. The drug appears to bind preferentially to mycobacterial DNA and inhibit mycobacterial replication and growth.
Binding of the drug to DNA appears to occur principally at base sequences containing guanine, with preferential binding to mycobacterial DNA resulting from the relatively high proportion of guanine and cytosine in this DNA compared with that in human DNA.
Clofazimine does not appear to undergo intercalative binding between base pairs of DNA. Clofazimine reportedly is slowly bactericidal against Mycobacterium leprae in vivo; however, evaluation of the effects of the drug on the organism is difficult since M. leprae cannot be cultured in vitro. Clofazimine is bactericidal against M. tuberculosis and M. marinum in vitro, but appears to be only bacteriostatic in vitro against other mycobacteria, including M. avium complex (MAC).
Anti-inflammatory and Immunosuppressive Effects
Clofazimine exerts anti-inflammatory and immunosuppressive effects in vitro and in vivo, although the precise mechanisms of these effects have not been fully elucidated. In vitro, clofazimine causes a progressive, dose-dependent inhibition of neutrophil motility and also inhibits mitogen-induced lymphocyte transformation. These effects also generally occurred in vivo in healthy adults and those with leprosy who received an oral clofazimine dosage of 200 mg daily for 5 days. In vitro, clofazimine increases both spontaneous and induced synthesis of prostaglandin E2 by polymorphonuclear leukocytes obtained from healthy individuals or patients with leprosy.
Although results of some studies are conflicting, most studies indicate that clofazimine enhances the phagocytic activity of polymorphonuclear cells and macrophages in vitro and in vivo and also enhances membrane-associated oxidative metabolism in these cells.
The anti-inflammatory and immunosuppressive effects of clofazimine, in addition to its antimycobacterial effects, apparently contribute to the efficacy of the drug in the treatment and prevention of erythema nodosum leprosum (ENL) reactions. Limited data, however, indicate that dapsone may decrease or nullify some of the anti-inflammatory effects of clofazimine and theoretically could adversely affect clofazimine’s efficacy in the treatment of ENL reactions. (See Drug Interactions: Dapsone.)
The mechanism of action of clofazimine in the treatment of inflammatory or pustular dermatosis (e.g., pyoderma gangrenosum) is unknown, but may be related to the drug’s enhancement of neutrophil and macrophage phagocytosis.
Spectrum Clofazimine is active against various Mycobacterium, including M. leprae, M. tuberculosis, M. avium complex (MAC), M. bovis, M. chelonei, M. fortuitum, M. kansasii, M. marinum, M. scrofulaceum, M. simiae, and M. ulcerans.
Although clofazimine has exhibited in vitro activity against some other bacteria, the drug is substantially less active against most of these bacteria than against mycobacteria and generally is not effective against these bacteria in vivo. The drug generally is inactive against other organisms, including fungi and protozoa. M. leprae cannot be cultured in vitro, but in vivo mouse footpad studies indicate that M. leprae generally is inhibited by clofazimine concentrations of 0.0001-0.001% in the diet.
Although mycobacterial killing may begin shortly after initiation of clofazimine therapy, it cannot be measured in biopsy tissues taken from patients for mouse footpad studies until approximately 50 days after the start of therapy.
Because clofazimine is highly hydrophobic, the drug must be dissolved in organic solvents (e.g., dimethyl sulfoxide, dioxane, methoxyethanol, or dimethylformamide) for use in in vitro susceptibility tests.
Agar or broth dilution susceptibility tests are generally preferred for determining the in vitro susceptibility of mycobacteria to clofazimine since poor diffusion of the drug into agar media may occur in in vitro disk-diffusion susceptibility tests. MAC isolates, including those obtained from patients with acquired immunodeficiency syndrome (AIDS), generally are inhibited in vitro by clofazimine concentrations of 0.1-10 mcg/mL. MICs of clofazimine for susceptible MAC strains generally are higher when determined using Lowenstein-Jensen media than when determined using Middlebrook 7H9, 7H10, or 7H11 media. In an in vitro study using Middlebrook 7H9 broth and MAC isolates from patients with AIDS, the MIC of clofazimine was 0.5-2 mcg/mL, but the MBC was greater than 32 mcg/mL. In vitro, M. tuberculosis, M. bovis, and M. ulcerans generally are inhibited by clofazimine concentrations of 0.01-3.3 mcg/mL, and M. kansasii and M. marinum are inhibited by clofazimine concentrations of 0.15-6.4 mcg/mL. In an in vitro study using cation-supplemented Mueller-Hinton agar, M. chelonei and M. fortuitum were inhibited by clofazimine concentrations of 0.25-8 mcg/mL; however, in another study using Middlebrook 7H9 broth, the MIC of the drug for these organisms was 32 mcg/mL or higher.
Several strains of an unidentified Mycobacterium sp. isolated from patients with Crohn’s disease were inhibited by clofazimine concentrations of 1-2 mcg/mL.
Resistance
Mycobacterium leprae resistant to clofazimine have been reported only rarely. In at least one patient, resistance to clofazimine was reported after 7 years of therapy with the drug; mouse footpad studies indicated that this isolate was resistant to clofazimine concentrations of 0.0001%, but was susceptible to concentrations of 0.001%. Cross-resistance between clofazimine and dapsone or rifampin has not been reported to date.
However, M. leprae resistant to both clofazimine and dapsone, but susceptible to rifampin, has been reported rarely.
Pharmacokinetics
Absorption
Clofazimine is incompletely absorbed from the GI tract following oral administration. The extent of oral absorption of the drug exhibits considerable interindividual variation and depends on several factors including particle size, dosage form administered, dosage, and presence of food in the GI tract.
About 20% of a dose is absorbed from the GI tract when clofazimine is administered as coarse crystals, but 45-70% of a dose may be absorbed when the drug is administered as capsules containing a microcrystalline (micronized) suspension of the drug in an oil-wax base. The fraction of the dose absorbed appears to decrease as oral dosage of clofazimine is increased. In a study in fasting lepromatous leprosy patients who received a single 100-, 300-, 400-, or 600-mg dose of clofazimine, approximately 62.5,48.7,44.3 or 42.6% of the dose, respectively, was absorbed.
Presence of food in the GI tract may increase the rate and extent of absorption of clofazimine. Results of a study in healthy adults indicate that the area under the serum concentration-time curve (AUC) of clofazimine is increased by about 60% and peak plasma concentrations are increased by about 30% when the drug is administered with food containing fat and protein, compared with the fasting state. In healthy adults who received a single 200-mg oral dose of clofazimine with food, peak plasma concentrations of the drug were attained 4-12 hours after the dose.
Results of multiple-dose studies indicate that steady-state plasma concentrations of clofazimine may not be attained until after at least 30 days of clofazimine therapy. In leprosy patients receiving clofazimine in a dosage of 100 or 300 mg once daily, steady-state serum concentrations of the drug generally average 0.7 or 1 mcg/mL, respectively. In a study in leprosy patients receiving 100 mg of the drug 3 times weekly, steady-state serum clofazimine concentrations averaged 0.5 mcg/mL.
In other leprosy patients receiving 100 mg of clofazimine 3 times weekly, steady-state serum concentrations of the drug have ranged from 0.05-0.4 mcg/mL. The relationship between plasma clofazimine concentrations and the therapeutic effects of the drug has not been determined.
Distribution
Clofazimine is highly lipophilic and is distributed principally to fatty tissue and cells of the reticuloendothelial system; the drug is taken up by macrophages throughout the body. In leprosy patients, clofazimine appears to distribute to and accumulate as crystals in highest concentrations in the mesenteric lymph nodes, adipose tissue, adrenals, liver, lungs, gallbladder, bile, and spleen and in lower concentrations in the skin, small intestine, lungs, heart, kidneys, pancreas, muscle, omentum, and bone.
Clofazimine crystals have also been found in bone marrow, sputum, sebum, and sweat, and in the iris, conjunctiva, macula, sclera, and cornea. The drug does not appear to distribute into the brain or CSF.
Clofazimine crosses the placenta and is distributed into milk.
Elimination
Following oral administration of a single dose of clofazimine in healthy adults, there is an initial distribution phase followed by a slow elimination phase with a terminal elimination half-life of approximately 8 days. The tissue half-life of clofazimine following multiple oral doses of the drug is estimated to be at least 70 days. The drug remains in body tissues for prolonged periods and is eliminated very slowly; clofazimine has been found in skin and in mesenteric lymph nodes 2 and 4 years, respectively, after therapy with the drug was discontinued. In mice infected with Mycobacterium leprae, once-daily and twice-weekly administration of the drug had comparable antileprotic activity.
The metabolic fate of clofazimine has not been fully elucidated, but the drug appears to accumulate in the body and to be excreted principally unchanged. Clofazimine appears to be partially metabolized and a least 3 metabolites have been found in urine of patients receiving the drug.
Metabolite I is formed by hydrolytic dehalogenation of clofazimine, metabolite II presumably is formed by a hydrolytic deamination reaction followed by glucuronidation, and metabolite III appears to be a hydrated clofazimine glucuronide. Clofazimine is excreted principally in feces, both as unabsorbed drug and via biliary elimination.
Fecal elimination of clofazimine exhibits considerable interindividual variation, and 35-74% of a single oral dose may be excreted unchanged in feces over the first 72 hours after the dose. Following oral administration of a single 200- or 300-mg dose, elimination of unchanged clofazimine and its metabolites in urine is negligible during the first 24 hours. Following multiple doses of the drug, less than 1% of the daily dose is excreted in urine over a 24-hour period. Small amounts of the drug also are excreted via sebaceous and sweat glands.
Chemistry and Stability
Chemistry
Clofazimine is a phenazine dye with antimycobacterial and anti-inflammatory activity. The dye is a substituted iminophenazine derivative and generally is described as bright red; however, the color varies depending on the degree of protonation, ranging from colorless (triprotonated) to orange (un-ionized or monoprotonated) or red (biprotonated). The drug is commercially available as capsules containing micronized clofazimine suspended in an oil-wax base. Clofazimine occurs as a fine, reddish brown powder. The drug is highly hydrophobic and is practically insoluble in water. The solubility of clofazimine in alcohol is 1 mg/mL at room temperature.
Stability
Clofazimine capsules should be stored in tight containers at 30°C or less and should be protected from moisture. However, USP states that the capsules can be stored in well-closed containers. Commercially available clofazimine capsules have an expiration date of 5 years following the date of manufacture.
Preparations
Clofazimine Oral Capsules 50 mg Lamprene®, (with parabens and propylene glycol) Novartis
Clofazimine: Side Effects
See also Antituberculosis drugs
Clofazimine is weakly bactericidal against Mycobacterium leprae. It is active in chronic skin ulcers (Buruli ulcer) and partly against Mycobacterium avium intracellulare. The usual adult dosage is 50-100 mg/day. At higher doses, its anti-inflammatory effect seems to prevent the development of acute reactions, such as erythema nodosum leprosum.
Clofazimine is a strongly lipophilic dye and accumulates in tissues, especially fat, bile, macrophages, the reticuloendothelial system, and skin. This is the basis of adverse reactions, including skin discoloration. Lymphedema, diminished sweating, and reduced tearing have been observed.
Observational studies
In 84 patients with leprosy who took clofazimine, the most common adverse effect was a dark red skin pigmentation of varying intensity, which appeared within 10 weeks of the start of therapy. The intensity of the colour was proportional to the density of the infiltration. There was ichthyosis in 56 patients. Adverse effects such as anorexia, diarrhea, lymph gland enlargement, liver enlargement, corneal drying, and loss of weight were self-correcting, but nine patients had severe gastrointestinal effects (severe abdominal pain, vomiting, and diarrhea).
Organs and Systems
Respiratory
Clofazimine crystals were observed in macrophages in the respiratory alveoli of a patient with AIDS; this is considered harmless.
Nervous system
Intraneural deposition of a ceroid-like pigment has been seen after treatment of lepromatous leprosy with clofazimine. This pigment does not affect the healing process. Treatment can be continued, provided that the dose is not too high.
Sensory systems
In 76 patients taking multidrug therapy, including clofazimine, for at least 6 months, 46% had conjunctival deposition, 53% had deposition in the cornea, and crystals were found in the tears of 32%. Conjunctival pigmentation has been reported, as well as reversible linear brownish corneal streaks. Two cases of macular pigmentation have also been described.
Gastrointestinal
Nausea, vomiting, diarrhea, abdominal pain, and anorexia have been reported with clofazimine.
Clofazimine can accumulate and precipitate in tissues, such as the wall of the small bowel, after prolonged administration. Enteropathy can develop if crystals are stored in the lamina propria of the jejunal mucosa and the mesenteric lymph nodes. These effects depend on the dosage and duration of therapy. When this complication is suspected jejunal biopsy is indicated. At laparotomy, all organs can have an orange-yellow color. On drug withdrawal, the enteropathy progressively improves. Recognized for the first time in 1967, only 14 cases of this complication have so far appeared in the English-language literature. Acute or chronic abdominal pain was the main symptom. In most patients, the diagnosis was made after exploratory laparotomy. Barium meal follow-through or CT scanning of the abdomen showed mucosal thickening in the small intestine. Mesenteric lymph node enlargement was present in the index case. Characteristic eosinophilic clofazimine crystals were demonstrated in the histiocytes of all patients except three. The authors proposed the term “clofazimine-induced crystal-storage histiocytosis” to emphasize causes other than B cell neoplasms for crystal-storage histiocytosis. Awareness of this complication of clofazimine can avoid unnecessary surgical exploration.
Liver
Clofazimine can inhibit the liver damage that is associated with lepromatous leprosy and the leprosy reaction; it has only minimal or no deleterious effects on liver function.
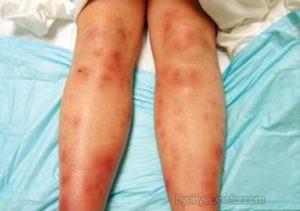
Skin
Pigmentation and purple skin discoloration are the most frequent adverse effects of clofazimine. It is therefore unacceptable to most light-skinned patients.
Ichthyosis is very frequent at dosages of clofazimine over 100 mg/day.
Nails
Nail changes, such as brown discoloration of the nail plate and onycholysis, have been described in patients taking high doses (300 mg/day) of clofazimine. Clofazimine crystals were demonstrated in the nails and nail beds.
Clofazimine: Cautions
Clofazimine generally is well tolerated when given in dosages of 100 mg daily or less. The major adverse effects of the drug involve the skin, eyes, and GI tract. Discoloration of various body tissues and fluids occurs in most patients receiving clofazimine, apparently because clofazimine is a bright-red dye and crystals of the drug are distributed to and accumulate in these tissues and fluids. Although most adverse effects of clofazimine are mild, dose related, and reversible following discontinuance of the drug, severe GI effects, which may be fatal, have been reported rarely.
Dermatologic Effects
Pink to brownish-black discoloration of the skin occurs in up to 75-100% of patients receiving clofazimine. The degree of skin discoloration is dose related and is most pronounced on exposed parts of the body, including the thin, hairless skin of the face (particularly periorbital and perinasal areas) and the thick, hairless, hypopigmented skin of the palms and soles, and in areas where leprosy lesions are located.
Occasionally, patients have described a feeling of tension in succulent, infiltrated lesions of the face accompanying the development of this discoloration. Biopsy of discolored skin has revealed the presence of granulomas, the extent of which appears to be related to the amount of drug deposited. Discoloration is most apparent in light-skinned individuals and may be particularly disturbing to these individuals.
Skin discoloration usually is evident 1-4 weeks after initiation of clofazimine therapy and gradually disappears within 6-12 months after discontinuance of the drug; however, the skin occasionally may retain traces of color for 4 years or longer. In addition to discoloration secondary to drug deposits, melanosis similar to that observed with phenothiazines has been reported in patients receiving clofazimine.
The blue-grey, blackish brown, to black discoloration associated with clofazimine-induced melanosis resolves more gradually following discontinuance of the drug and may persist as circumscribed hyperpigmented areas in some patients.
During therapy with the drug, leprosy nodules may be replaced by scar tissue in the form of shiny, jet black, circular macules. Ichthyosis and dry skin, especially on the legs and forearms, have been reported in 8-38% of patients receiving clofazimine. Clofazimine-induced ichthyosis and dryness generally occur as leprosy resolves and may be relieved by applying oil, petrolatum, or an emollient lotion containing 25% urea to the affected areas.
Desquamation may occur. Pruritus and nonspecific rash (e.g., follicular and papular) have been reported in 1-5%1 and seborrheic dermatitis, erythroderma, erythema multiforme, acneiform eruptions, monilial cheilosis (perleche), and phototoxicity have been reported in less than 1% of patients receiving clofazimine.
Severe exfoliative dermatitis, which appeared to be a hypersensitivity reaction, occurred in at least one patient receiving the drug.
Ocular Effects
Reversible, dose-related, red-brown discoloration of the conjunctiva, cornea, and lacrimal fluid may occur during clofazimine therapy. Conjunctival discoloration has been reported in 38-57% of patients receiving the drug. Bilateral, linear or branched, brownish lines or streaks in the cornea have been reported in some patients receiving clofazimine dosages of 100-400 mg daily for 2 months or longer; these lines slowly disappeared after clofazimine therapy was completed.
Discoloration in the macular areas of the eye and bluish discoloration of the lens have also been reported rarely. Although discoloration of the conjunctiva and other parts of the eye during clofazimine therapy does not appear to affect visual acuity, diminished vision has been reported rarely in patients receiving the drug. Dryness, burning, itching, irritation, and watering of the eye have been reported in 2-32% of patients receiving clofazimine.
GI Effects
GI effects are the major dose-limiting adverse effects of clofazimine. Abdominal and epigastric pain, diarrhea, nausea, vomiting, and GI intolerance have been reported in up to 60% of patients receiving the drug.
Anorexia, weight loss, malabsorption, constipation, bowel congestion or obstruction, GI bleeding, splenic infarction, and eosinophilic enteritis have been reported in less than 1% of patients receiving the drug.
Adverse GI effects appear to be dose related and occur most frequently in patients receiving more than 100 mg of clofazimine daily. In patients who receive usual dosages of the drug, adverse GI effects may occur within a few days or weeks after initiation of clofazimine therapy and apparently are caused by a direct irritant effect of the drug on the intestinal mucosa; GI symptoms in these patients generally subside following a decrease in clofazimine dosage. In patients who receive 300 mg or more of the drug daily, a severe syndrome of crampy or colicky abdominal pain, persistent diarrhea, and weight loss may occur 3 months or longer after initiation of clofazimine therapy; discontinuance of the drug usually is, and hospitalization may be, necessary.
If clofazimine therapy is not discontinued, partial or complete bowel obstruction may occur. Severe abdominal symptoms have necessitated exploratory laparotomies in some patients and several fatalities have been reported. Although the exact cause of this syndrome is unknown, massive deposits of clofazimine crystals in various tissues including the intestinal mucosa, liver, spleen, and mesenteric lymph nodes have been observed.
Nervous System Effects
Dizziness, drowsiness, fatigue, headache, giddiness, neuralgia, and taste disorder have been reported in less than 1% of patients receiving clofazimine. Depression, secondary to skin discoloration, may occur in some patients receiving clofazimine; this may have contributed to at least 2 suicides in patients receiving the drug.
Other Effects
Reversible, dose-related, red-brown discoloration of sweat, sputum, urine, feces, nasal secretions, semen, and breast milk may occur during clofazimine therapy. Although a causal relationship has not been established, increased blood glucose concentrations have been reported in more than 1% of patients receiving the drug.
Eosinophilia, anemia, hypokalemia, elevated erythrocyte sedimentation rate (ESR), and elevated concentrations of serum albumin, bilirubin, and AST (SGOT) have been reported in less than 1% of patients receiving clofazimine. Thromboembolism, cystitis, bone pain, edema, fever, lymphadenopathy, vascular pain, hepatitis, jaundice, and enlarged liver also have been reported in less than 1% of patients receiving the drug.
Precautions and Contraindications
Clofazimine dosages exceeding 100 mg daily should be used for as short a period as possible and only under close medical supervision. Severe GI effects, including splenic infarction, bowel obstruction, and GI bleeding, have been reported rarely during clofazimine therapy and have been fatal in a few patients.
Clofazimine should be used with caution in patients with GI symptoms such as abdominal pain and diarrhea. If a patient complains of colicky or burning abdominal pain, nausea, vomiting, or diarrhea during clofazimine therapy, dosage of the drug should be reduced and, if necessary, the interval between doses increased, or the drug should be discontinued.
Patients receiving clofazimine should be warned that the drug may cause a pink to brownish-black discoloration of the skin as well as discoloration of the conjunctiva, lacrimal fluid, sweat, sputum, urine, feces, nasal secretions, semen, and breast milk.
Patients should be advised that skin discoloration, although reversible, may take several months or years to disappear after completion of clofazimine therapy. The fact that skin discoloration may result in mental depression in some patients should be considered; depression secondary to skin discoloration may have contributed to at least 2 suicides in patients receiving clofazimine. The manufacturer states that there are no known contraindications to use of clofazimine.
Pediatric Precautions
Safety and efficacy of clofazimine in children 12 years of age or younger have not been established. The drug has been used in a limited number of children.
Mutagenicity and Carcinogenicity
Clofazimine was not mutagenic in the Ames microbial mutagen test with or without metabolic activation. Studies have not been performed to date to evaluate the carcinogenic potential of clofazimine.
Pregnancy, Fertitlity and Lactation
Clofazimine did not appear to be teratogenic in rabbits or rats when given at dosages 8 or 25 times the usual human dosage, respectively. However, in reproduction studies in mice using clofazimine dosages 12-25 times the usual human dosage, the drug caused retardation of fetal skull ossification, an increased incidence of abortions and stillbirths, and impaired neonatal survival. There are no adequate and controlled studies to date using clofazimine in pregnant women, and the drug should be used during pregnancy only when the potential benefits justify the possible risks to the fetus.
Clofazimine has been used during (including throughout) pregnancy in a limited number of women without evidence of teratogenicity. However, clofazimine does cross the placenta, and the skin of some neonates born to women who received the drug during pregnancy was discolored at birth; the discoloration gradually faded over the first year. Several neonates born to women receiving the drug during pregnancy have died, but a causal relationship to clofazimine has not been established.
Although further study is needed, there is some evidence that estrogen excretion may be decreased in pregnant women receiving clofazimine. In reproduction studies in rats receiving a clofazimine dosage 25 times the usual human dosage, there was some evidence of impaired fertility since the number of offspring was reduced and there was a lower proportion of implantations.
Because clofazimine is distributed into milk, the drug should not be used in nursing women unless clearly indicated since skin discoloration can occur in nursing infants of such women.
| Dosage forms of Clofazimine: |
|---|
| Clofazimine powder |
Synonyms of Clofazimine:
Chlofazimine, Clofazimina [INN-Spanish], Clofaziminum [INN-Latin]
How can i get Clofazimine online over the counter?
You can buy Clofazimine OTC in online drugstore with low cost.
Therapeutic classes of Clofazimine:
Antimycobacterials, Coloring Agents, Dyes, Leprostatic Agents
Delivery
Australia, Canada, Mexico, New Zealand, USA, Europe [Belgium, France, Norway, Holland, Ireland, Spain, Switzerland, Great Britain (UK), Italy] and etc.


 (10 votes, average: 4.00 out of 5)
(10 votes, average: 4.00 out of 5)
Hi. I want to buy caps lamprene 100 mg. I am in Greece. Please sent my an answer as quickly as possible. Thank you.